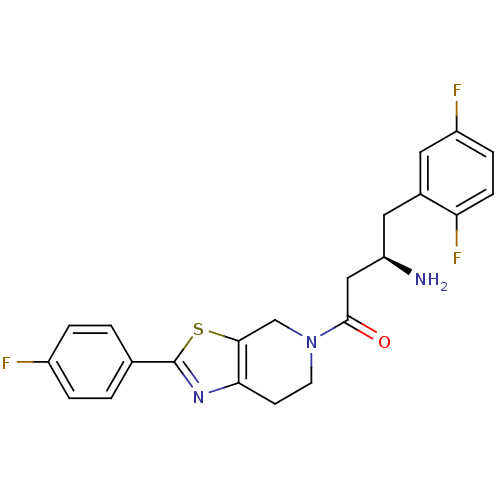
BDBM11966 (3R)-3-amino-4-(2,5-difluorophenyl)-1-[2-(4-fluorophenyl)-4H,5H,6H,7H-pyrido[4,3-d][1,3]thiazol-5-yl]butan-1-one::thiazolopiperidine 68
SMILES N[C@@H](CC(=O)N1CCc2nc(sc2C1)-c1ccc(F)cc1)Cc1cc(F)ccc1F
InChI Key InChIKey=LRGMUQFAJNIPGS-UHFFFAOYSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 11966
Found 4 hits for monomerid = 11966
Affinity DataIC50: 360nMAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 4.30E+3nMAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 7.80E+3nMAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair