BDBM158505 US9034854, E4::US9040509, E4::US9561238, E4
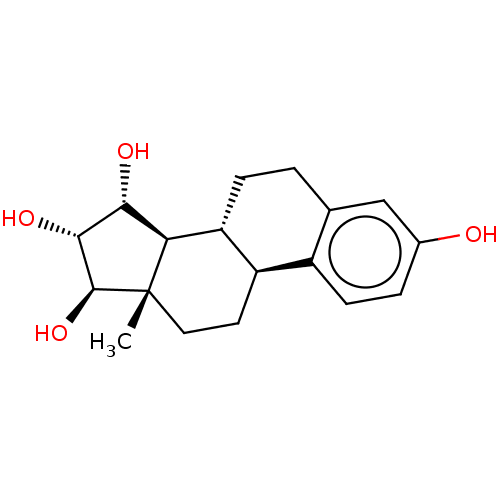
SMILES C[C@]12CC[C@@H]3c4ccc(cc4CC[C@H]3[C@@H]1[C@H]([C@H]([C@@H]2O)O)O)O
InChI Key InChIKey=AJIPIJNNOJSSQC-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 158505
Found 6 hits for monomerid = 158505
Affinity DataKi: 4.90nMpH: 7.5Assay Description:The method employed was adapted from the scientific literature and described in detail by Osbourn et al. (1993, Biochemistry, 32, 6229-6236). Recombi...More data for this Ligand-Target Pair
Affinity DataKi: 4.90nM ΔG°: -11.3kcal/molepH: 7.5 T: 2°CAssay Description:The method employed was adapted from the scientific literature and described in detail by Osboum et al. (1993, Biochemistry, 32, 6229-6236). Recombin...More data for this Ligand-Target Pair
Affinity DataKi: 4.90nM ΔG°: -11.3kcal/molepH: 7.5 T: 2°CAssay Description:The method employed was adapted from the scientific literature and described in detail by Osbourn et al. (1993, Biochemistry, 32, 6229-6236). Recombi...More data for this Ligand-Target Pair
Affinity DataKi: 19nM ΔG°: -10.5kcal/molepH: 7.5 T: 2°CAssay Description:The method employed was adapted from the scientific literature and described in detail by Osboum et al. (1993, Biochemistry, 32, 6229-6236). Recombin...More data for this Ligand-Target Pair
Affinity DataKi: 19nMpH: 7.5Assay Description:The method employed was adapted from the scientific literature and described in detail by Osbourn et al. (1993, Biochemistry, 32, 6229-6236). Recombi...More data for this Ligand-Target Pair
Affinity DataKi: 19nM ΔG°: -10.5kcal/molepH: 7.5 T: 2°CAssay Description:The method employed was adapted from the scientific literature and described in detail by Osbourn et al. (1993, Biochemistry, 32, 6229-6236). Recombi...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)