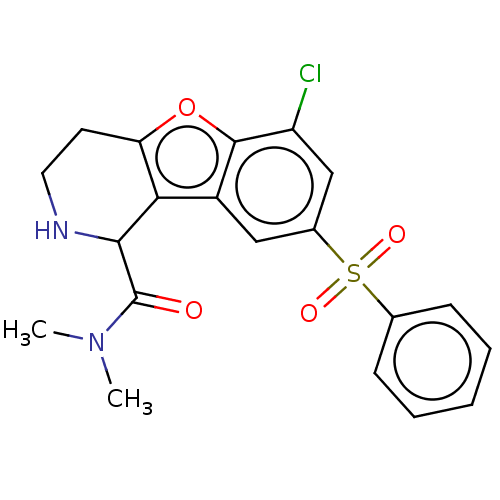
BDBM166231 US9067949, 88
SMILES CN(C)C(=O)C1NCCc2oc3c(Cl)cc(cc3c12)S(=O)(=O)c1ccccc1
InChI Key InChIKey=HEXOMDSSQWRBOQ-UHFFFAOYSA-N
Data 1 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 1 hit for monomerid = 166231
Found 1 hit for monomerid = 166231
Affinity DataKi: 252nM ΔG°: -9.00kcal/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair