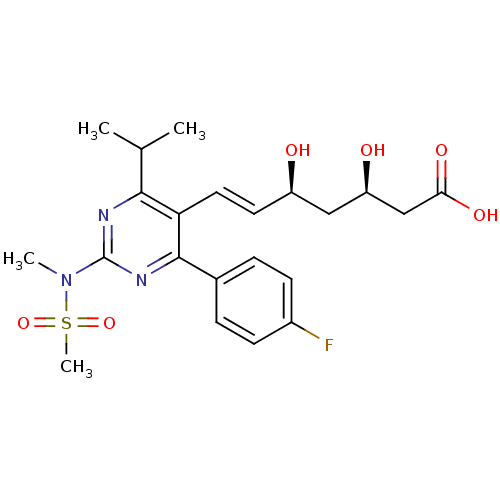
BDBM18372 (3R,5S,6E)-7-[4-(4-fluorophenyl)-2-(N-methylmethanesulfonamido)-6-(propan-2-yl)pyrimidin-5-yl]-3,5-dihydroxyhept-6-enoic acid::CHEMBL1496::Ros::Rosuvastatin::US9102656, Rosuvastatin ::ZD4522
SMILES CC(C)c1nc(nc(-c2ccc(F)cc2)c1\C=C\[C@@H](O)C[C@@H](O)CC(O)=O)N(C)S(C)(=O)=O
InChI Key InChIKey=BPRHUIZQVSMCRT-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 15 hits for monomerid = 18372
Found 15 hits for monomerid = 18372
Affinity DataIC50: 3.10nM EC50: 0.300nMpH: 7.2 T: 2°CAssay Description:Enzyme Assay for HMG-CoA reductase was based on the conversion of isotopically labeled HMG-CoA to mevalonic acid using rat liver microsomes as enzyme...More data for this Ligand-Target Pair
Affinity DataIC50: 3.10nM EC50: 0.600nMpH: 7.0 T: 2°CAssay Description:Enzyme Assay for HMG-CoA reductase was based on the conversion of isotopically labeled HMG-CoA to mevalonic acid using rat liver microsomes as enzyme...More data for this Ligand-Target Pair
Target3-hydroxy-3-methylglutaryl-coenzyme A reductase(Human)
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Affinity DataKi: 0.900nMAssay Description:Inhibitory constant against HMG-CoA reductaseMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMAssay Description:Assay for HMG-CoA reductase was based on the conversion of isotopically labeled HMG-CoA to mevalonic acid using rat liver microsomes as enzyme source...More data for this Ligand-Target Pair
Target3-hydroxy-3-methylglutaryl-coenzyme A reductase(Human)
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Affinity DataIC50: 3.10nMAssay Description:Inhibition of HMG-CoA reductaseMore data for this Ligand-Target Pair
Affinity DataIC50: 3.10nMAssay Description:Inhibition of HMG-CoA reductase in Sprague-Dawley rat liver microsomes using [14C]HMG-CoA as substrate preincubated for 0.5 hrs before substrate addi...More data for this Ligand-Target Pair
Affinity DataIC50: 3.60nMpH: 7.2 T: 2°CAssay Description:Assay for HMG-CoA reductase was based on the conversion of isotopically labeled HMG-CoA to mevalonic acid using rat liver microsomes as enzyme source...More data for this Ligand-Target Pair
Affinity DataIC50: 3.70nMAssay Description:Inhibition of rat microsomal HMGCoA reductaseMore data for this Ligand-Target Pair
Target3-hydroxy-3-methylglutaryl-coenzyme A reductase(Human)
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:The following procedure was followed using a HMG-CoA Reductase assay kit obtained from Sigma-Aldrich (catalogue number CS1090). The assay is based on...More data for this Ligand-Target Pair
Target3-hydroxy-3-methylglutaryl-coenzyme A reductase(Human)
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:Inhibitory concentration against 3-hydroxy-3-methylglutaryl-CoA reductaseMore data for this Ligand-Target Pair
Target3-hydroxy-3-methylglutaryl-coenzyme A reductase(Human)
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Affinity DataIC50: 5.40nMAssay Description:Inhibitory concentration against HMG-CoA reductaseMore data for this Ligand-Target Pair
TargetRetinal rod rhodopsin-sensitive cGMP 3',5'-cyclic phosphodiesterase subunit delta(Human)
Max Planck Institute of Molecular Physiology
Curated by ChEMBL
Max Planck Institute of Molecular Physiology
Curated by ChEMBL
Affinity DataKd: 1.25E+3nMAssay Description:Binding affinity to His6-tagged PDE-delta (unknown origin) measured every 3 mins by fluorescence polarization assayMore data for this Ligand-Target Pair
Target3-hydroxy-3-methylglutaryl-coenzyme A reductase(Human)
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Universidad Nacional AutóNoma De MéXico
Curated by ChEMBL
Affinity DataKi: 7.10E+4nMAssay Description:Inhibitory constant against HMG-CoA reductase with alpha asaroneMore data for this Ligand-Target Pair
Activity Spreadsheet -- ITC Data from BindingDB
 Found 1 hit for monomerid = 18372
Found 1 hit for monomerid = 18372
Cell3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMG-CoA)(Human)
Boehringer Ingelheim Pharmaceuticals
Boehringer Ingelheim Pharmaceuticals
ITC DataΔG°: -10.8kcal/mole −TΔS°: 2.40kcal/mole ΔH°: -13.2kcal/mole logk: 5.88E+7
pH: 7.2 T: 30.00°C
pH: 7.2 T: 30.00°C