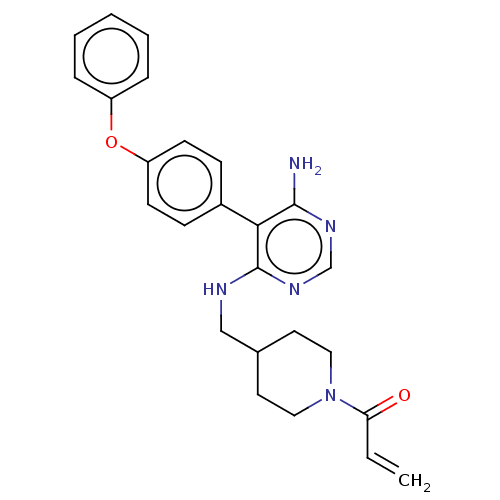
BDBM291522 1-(4-(((6-amino-5-(4-phenoxyphenyl)pyrimidin-4-yl)amino)methyl)piperidin-1-yl)prop-2-en-1-one ::US10413562, Compound A250::US9580449, Example A250::US9580449, Example A39
SMILES Nc1ncnc(NCC2CCN(CC2)C(=O)C=C)c1-c1ccc(Oc2ccccc2)cc1
InChI Key InChIKey=QUIWHXQETADMGN-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 34 hits for monomerid = 291522
Found 34 hits for monomerid = 291522
Affinity DataIC50: 1.80nMAssay Description:Inhibition of BTK (unknown origin) measured by ADP-Glo assayMore data for this Ligand-Target Pair
Affinity DataKd: 4.5nMAssay Description:Binding affinity to DNA-tagged recombinant TEC (unknown origin) measured after 1 hr by biotinylated-ligand affinity bead-based qPCR analysisMore data for this Ligand-Target Pair
Affinity DataKd: 4.60nMAssay Description:Binding affinity to wild-type human full length BTK (M1 to S659 residues) expressed in mammalian expression system by Kinomescan methodMore data for this Ligand-Target Pair
Affinity DataKd: 4.60nMAssay Description:Inhibition of human full-length wild type BTK (M1 to S659 residues) expressed in mammalian expression systemMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of human BTK using fluorescein-labeled polyGAT peptide as substrate incubated for 30 mins by FRET assayMore data for this Ligand-Target Pair
Affinity DataIC50: 8.90nMAssay Description:Inhibition of BTK (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 8.90nMAssay Description:Covalent inhibition of N-terminal GST-fused human BTK (2-659(end) amino acids) expressed in baculovirus expression system using FITC-AHA-EEPLYWSFPAKK...More data for this Ligand-Target Pair
Affinity DataKd: 16nMAssay Description:Binding affinity to DNA-tagged recombinant BTK (unknown origin) measured after 1 hr by biotinylated-ligand affinity bead-based qPCR analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Inhibition of BTK (unknown origin) incubated for 140 mins in presence of ATP and [gamma-33p] ATP by hotspot assayMore data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Inhibition of FLAG-tagged TEC autophosphorylation in HEK293 cells incubated for 2 hrs by MSD electrochemiluminescence immunoassayMore data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Inhibition of BTK phosphorylation at Tyr 223 residue in human Ramos cell incubated for 3 hrs by HTRF-based analysisMore data for this Ligand-Target Pair
Affinity DataKd: 31nMAssay Description:Binding affinity to DNA-tagged recombinant BMX (unknown origin) measured after 1 hr by biotinylated-ligand affinity bead-based qPCR analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:Inhibition of cytoplasmic recombinant human full length His-tagged BMX expressed in baculovirus by Z'-LYTE assayMore data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:Inhibition of recombinant human cytoplasmic GST-tagged ERBB4 expressed in baculovirus by Z'-LYTE assayMore data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:Inhibition of recombinant human cytoplasmic full length His-tagged TEC expressed in baculovirus by Z'-LYTE assayMore data for this Ligand-Target Pair
Affinity DataIC50: 61nMAssay Description:Inhibition of BTK in human PBMC cells assessed as reduction in anti-IgM-stimulated CD69 expression on B cells preincubated for 60 mins followed by go...More data for this Ligand-Target Pair
Affinity DataIC50: 61nMAssay Description:Inhibition of BTK in vitamin D3 differentiated human THP1 cells assessed as inhibition of FCgammaR-induced IL8 production measured after 24 hrs by HT...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:Inhibition of full-length human recombinant BTK using FITC-Ahx-TSELKKVVALYDYMPMNAND-NH2 as substrate measured after 60 mins by caliper assayMore data for this Ligand-Target Pair
Affinity DataIC50: 100nMpH: 7.5 T: 2°CAssay Description:The following describes a microfluidic, off-chip mobility shift kinase assay used to measure inherent potency of compounds against BTK enzyme. Compou...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:2.5× stocks of full-length human BTK (08-080) from CarnaBio USA, Inc., Natick, Mass., 1.6×ATP and appropriate kinKDR peptide substrate (FITC-AHA-EEPL...More data for this Ligand-Target Pair
Affinity DataIC50: 320nMAssay Description:Inhibition of BTK in human B cells assessed as reduction in anti-IgM/IL4-stimulated CD69 expression on B cells preincubated for 60 mins followed by a...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 7.5 T: 2°CAssay Description:The following describes a microfluidic, off-chip mobility shift kinase assay used to measure inherent potency of compounds against BTK enzyme. Compou...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of CD69 in human whole blood preincubated for 30 mins followed by anti-human IgD stimulation and measured after 18 to 22 hrs by flow cytom...More data for this Ligand-Target Pair
Affinity DataIC50: 1.18E+3nMAssay Description:Inhibition of BTK in human basophils assessed as reduction in anti-IgE mouse IgG1 antibody Le2-stimulated CD63 expression on basophil preincubated fo...More data for this Ligand-Target Pair
Affinity DataKi: 3.10E+3nMAssay Description:Inhibition of human ERG expressed in HEK293 cells at -80 mV holding potential by HPLC analysisMore data for this Ligand-Target Pair
Affinity DataKd: 3.70E+3nMAssay Description:Binding affinity to DNA-tagged recombinant ITK (unknown origin) measured after 1 hr by biotinylated-ligand affinity bead-based qPCR analysisMore data for this Ligand-Target Pair
Affinity DataKd: 5.60E+3nMAssay Description:Binding affinity to DNA-tagged recombinant ERBB4 (unknown origin) measured after 1 hr by biotinylated-ligand affinity bead-based qPCR analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 5.80E+3nMAssay Description:Inhibition of N-terminal GST-tagged human EGFR (696 to end aminoacids) expressed in baculovirus infected Sf21 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 5.84E+3nMAssay Description:Inhibition of GP6 in human whole blood assessed as protein-mediated platelet aggregation preincubated for 15 mins followed by collagen stimulation an...More data for this Ligand-Target Pair
Affinity DataKd: 7.10E+3nMAssay Description:Binding affinity to DNA-tagged recombinant EGFR (unknown origin) measured after 1 hr by biotinylated-ligand affinity bead-based qPCR analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 8.60E+3nMAssay Description:Inhibition of BTK Cys481S mutant (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of EGFR autophosphorylation at Tyr1068 residue in EGFR-amplified human A-431 cells preincubated with compound for 1 hrs followed by stimul...More data for this Ligand-Target Pair
Affinity DataKd: >1.00E+4nMAssay Description:Binding affinity to DNA-tagged recombinant ERBB2 (unknown origin) measured after 1 hr by biotinylated-ligand affinity bead-based qPCR analysisMore data for this Ligand-Target Pair
Affinity DataKd: >1.00E+4nMAssay Description:Binding affinity to DNA-tagged recombinant JAK3 (unknown origin) measured after 1 hr by biotinylated-ligand affinity bead-based qPCR analysisMore data for this Ligand-Target Pair