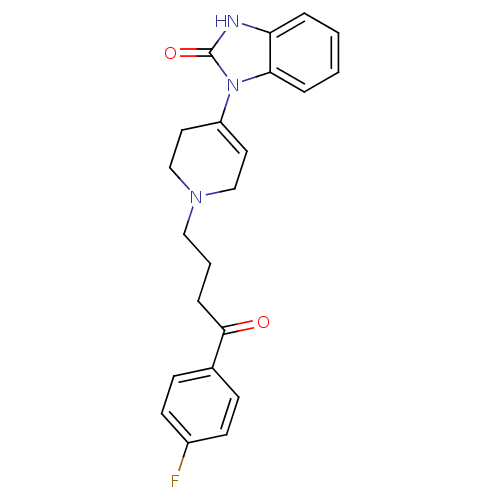
BDBM50017705 1-(1-(4-(4-fluorophenyl)-4-oxobutyl)-1,2,3,6-tetrahydropyridin-4-yl)-1H-benzo[d]imidazol-2(3H)-one::1-{1-[4-(4-Chloro-phenyl)-4-oxo-butyl]-1,2,3,6-tetrahydro-pyridin-4-yl}-1,3-dihydro-benzoimidazol-2-one(Droperidol)::1-{1-[4-(4-Fluoro-phenyl)-4-oxo-butyl]-1,2,3,6-tetrahydro-pyridin-4-yl}-1,3-dihydro-benzoimidazol-2-one::1-{1-[4-(4-fluorophenyl)-4-oxobutyl]-1,2,3,6-tetrahydro-4-pyridinyl}-2,3-dihydro-1H-benzo[d]imidazol-2-one::CHEMBL1108::DROPERIDOL::Inapsine::McN-JR-4749::R-4749
SMILES c1ccc2c(c1)NC(=O)N2C3=CCN(CC3)CCCC(=O)c4ccc(cc4)F
InChI Key InChIKey=RMEDXOLNCUSCGS-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 18 hits for monomerid = 50017705
Found 18 hits for monomerid = 50017705
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:Inhibition of human voltage-gated potassium channel subunit Kv11.1 (ERG K+ channel) in open stateMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:Inhibition of human Potassium channel HERG expressed in mammalian cellsMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 32.2nMAssay Description:K+ channel blocking activity in human embryonic kidney cells expressing HERG Kv11.1More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 32.4nMAssay Description:Inhibitory concentration against potassium channel HERGMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 32.4nMAssay Description:Inhibition of human ERG expressed in CHO cells by whole cell patch clamp techniqueMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 32.4nMAssay Description:Inhibition of human ERGMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 32.4nMAssay Description:Inhibition of human ERG channel in HEK293 cells by voltage-clamp methodMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 32.4nMAssay Description:Inhibition of human ERG in MCF7 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 740nMAssay Description:Inhibition of binding of Batrachotoxinin [3H]BTX-B to high affinity sites on voltage dependent sodium channels in a vesicular preparation from guinea...More data for this Ligand-Target Pair
TargetVoltage-dependent L-type calcium channel subunit alpha-1C/alpha-1D/alpha-1F/alpha-1S(Human)
Chantest
Curated by ChEMBL
Chantest
Curated by ChEMBL
Affinity DataIC50: 7.60E+3nMAssay Description:Inhibition of Cav1.2 current measured using QPatch automatic path clamp system in CHO cells expressing Cav1.2, beta-2 and alpha-2/delta-1 subunitsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human BSEP expressed in baculovirus transfected fall armyworm Sf21 cell membranes vesicles assessed as reduction in ATP-dependent [3H]-...More data for this Ligand-Target Pair
TargetN-acetyltransferase Eis(Mycobacterium tuberculosis (strain ATCC 25618 / H3...)
University of Kentucky
Curated by ChEMBL
University of Kentucky
Curated by ChEMBL
Affinity DataIC50: 1.22E+4nMAssay Description:Inhibition of Mycobacterium tuberculosis Eis assessed as Eis-mediated kanamycin acetylation preincubated for 10 mins followed by substrate addition a...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)