BDBM50032625 CHEMBL3354187
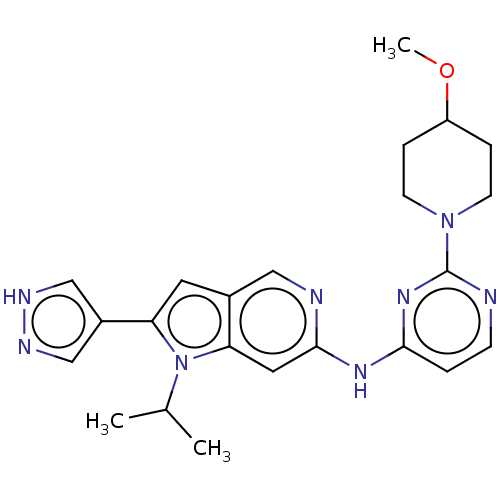
SMILES COC1CCN(CC1)c1nccc(Nc2cc3n(C(C)C)c(cc3cn2)-c2cn[nH]c2)n1
InChI Key InChIKey=GZCREURYZWXRHS-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50032625
Found 6 hits for monomerid = 50032625
Affinity DataKi: 16nMAssay Description:Inhibition of EGFR T790M/L858R mutant (unknown origin) by high-throughput biochemical screeningMore data for this Ligand-Target Pair
Affinity DataKi: 367nMAssay Description:Inhibition of EGFR T790M/del746 to 750 mutant (unknown origin) by high-throughput biochemical screeningMore data for this Ligand-Target Pair
Affinity DataIC50: 154nMAssay Description:Inhibition of Jak2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Inhibition of KDR (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 107nMAssay Description:Inhibition of Aurora B (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of CDK2 (unknown origin)More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)