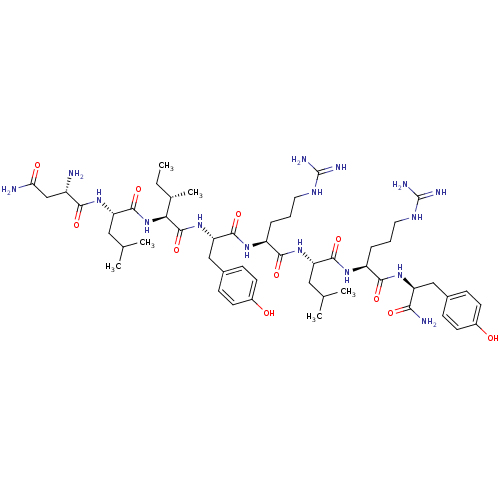
BDBM50034120 Asn-Leu-Ile-Tyr-Arg-Leu-Arg-Tyr-NH2::CHEMBL428097
SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CC(N)=O)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O
InChI Key InChIKey=PXAANFHGWQBXQR-ZSBWQBSESA-N
Data 1 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 1 hit for monomerid = 50034120
Found 1 hit for monomerid = 50034120
Affinity DataIC50: 310nMAssay Description:50% displacement of specifically bound [3H]-NPY2 from rat brain membranes.More data for this Ligand-Target Pair