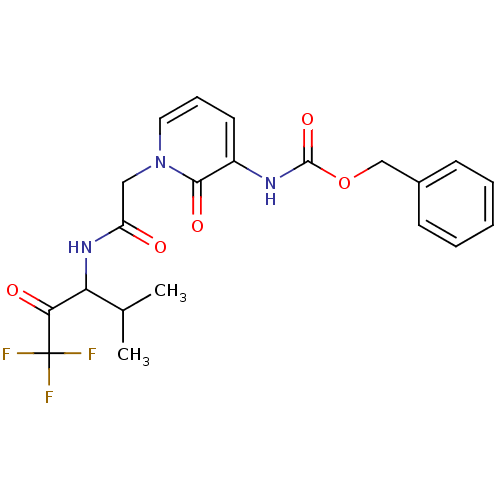
BDBM50037109 CHEMBL13684::{2-Oxo-1-[(3,3,3-trifluoro-1-isopropyl-2-oxo-propylcarbamoyl)-methyl]-1,2-dihydro-pyridin-3-yl}-carbamic acid benzyl ester
SMILES CC(C)C(NC(=O)Cn1cccc(NC(=O)OCc2ccccc2)c1=O)C(=O)C(F)(F)F
InChI Key InChIKey=KDWLXRNZUBXOCU-UHFFFAOYSA-N
Data 5 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50037109
Found 5 hits for monomerid = 50037109
Affinity DataKi: 280nMAssay Description:Inhibition of Human Neutrophil Elastase-catalyzed hydrolysis of the synthetic substrate MeO-Suc-Ala-Ala-Pro-Val-p- nitroanilide.More data for this Ligand-Target Pair
Affinity DataKi: 280nMAssay Description:The compound was tested in vitro for ability to inhibit human leukocyte elastase activityMore data for this Ligand-Target Pair
Affinity DataKi: 280nMAssay Description:Tested for inhibitory activity against human leukocyte elastase(HLE) enzymeMore data for this Ligand-Target Pair
Affinity DataKi: 280nMAssay Description:Inhibition of Human Neutrophil Elastase-catalyzed hydrolysis of the synthetic substrate MeO-Suc-Ala-Ala-Pro-Val-p- nitroanilide.More data for this Ligand-Target Pair
Affinity DataKi: 3.30E+5nMAssay Description:Tested for inhibitory activity against bovine pancreatic chymotrypsinogenMore data for this Ligand-Target Pair