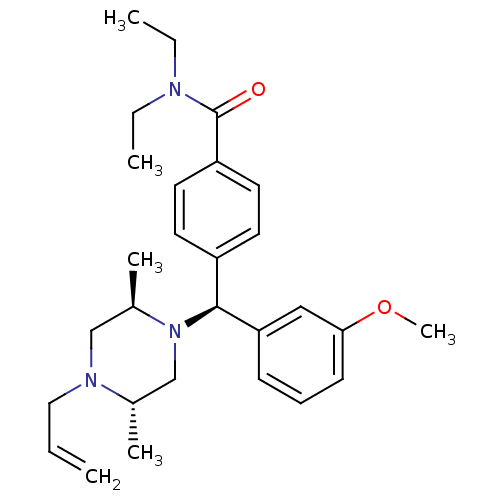
BDBM50039024 4-[(S)-((2R,5S)-4-Allyl-2,5-dimethyl-piperazin-1-yl)-(3-methoxy-phenyl)-methyl]-N,N-diethyl-benzamide::CHEMBL65648::cid_16219954
SMILES CCN(CC)C(=O)c1ccc(cc1)[C@H](N1C[C@H](C)N(CC=C)C[C@H]1C)c1cccc(OC)c1
InChI Key InChIKey=KQWVAUSXZDRQPZ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 50039024
Found 13 hits for monomerid = 50039024
TargetDelta-type opioid receptor(Mouse)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Tested for inhibition of binding of [3H]DADLE to mouse brain membranes depleted of mu binding sites by pretreatment with irreversible ligand BIT for ...More data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Mouse)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 2.70nMAssay Description:Tested for agonist activity against delta opioid receptor mouse vas deferensMore data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Human)
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
Affinity DataEC50: 10.7nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Affiliation: The Scripps Research Institute, TSRI Assa...More data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Mouse)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 31nMAssay Description:Agonist activity was evaluated in mouse vas deferens (MVD) preparations at Opioid receptor delta 1More data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Rat)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 63nMAssay Description:Inhibition of radioligand [3H]DADLE binding to rat brain Opioid receptor delta 1More data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Rat)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 63nMAssay Description:Binding affinity for Opioid receptor delta 1 by the inhibition of binding of [3H]DADAL in rat brain membranes.More data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Rat)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 63.3nMAssay Description:Inhibition of radioligand [3H]DADLE binding to rat brain Opioid receptor delta 1More data for this Ligand-Target Pair
TargetMu-type opioid receptor(Guinea pig)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 1.52E+3nMAssay Description:Agonist activity was evaluated in guinea pig ileum (GPI) preparations at Opioid receptor mu 1More data for this Ligand-Target Pair
TargetMu-type opioid receptor(Rat)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 2.47E+3nMAssay Description:Evaluated for inhibition of [3H]DAMGO binding from mu receptor in rat brain membranesMore data for this Ligand-Target Pair
TargetMu-type opioid receptor(Guinea pig)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 5.46E+3nMAssay Description:Tested for agonist activity against mu opioid receptor in guinea pig ileumMore data for this Ligand-Target Pair
TargetMu-type opioid receptor(Rat)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 5.71E+3nMAssay Description:Inhibition of radioligand [3H]DAMGO binding to rat brain Opioid receptor mu 1 using 100 nM DAMGOMore data for this Ligand-Target Pair
TargetMu-type opioid receptor(Rat)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 5.71E+3nMAssay Description:Binding affinity for Opioid receptor mu 1 by the inhibition of binding of [3H]DAMGO in rat brain membranes.More data for this Ligand-Target Pair
TargetMu-type opioid receptor(Rat)
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataIC50: 5.71E+3nMAssay Description:Inhibition of radioligand [3H]DAMGO binding to rat brain Opioid receptor mu 1 using 100 nM DAMGOMore data for this Ligand-Target Pair