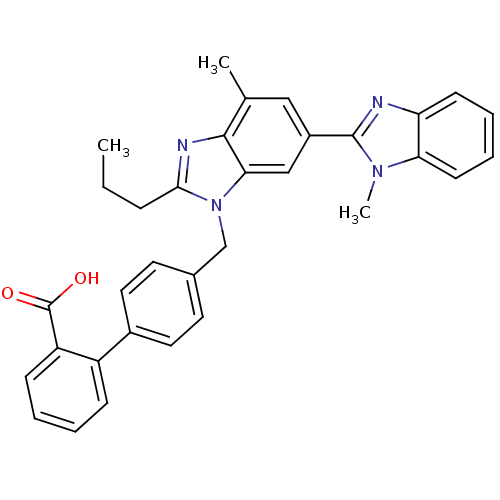
BDBM50043280 4'-((1,4'-dimethyl-2'-propyl(2,6'-bi-1H-benzimidazol)-1'-yl)methyl)-(1,1'-biphenyl)-2-carboxylic acid::4'-((4-methyl-6-(1-methyl-2-benzimidazolyl)-2-propyl-1-benzimidazolyl)methyl)-2-biphenylcarboxylic acid::4'-[(1,4'-dimethyl-2'propyl[2,6'-bi-1H-benzimidazol]-1'-yl)methyl]-[1,1'-biphenyl]-2-carboxylic acid::4'-[(1,7'-dimethyl-2'-propyl-1H,3'H-2,5'-bibenzimidazol-3'-yl)methyl][1,1'-biphenyl]-2-carboxylic acid::4'-[(1,7'-dimethyl-2'-propyl-1H,3'H-2,5'-bibenzimidazol-3'-yl)methyl]biphenyl-2-carboxylic acid::BIBR 277::CHEMBL1017::TELMISARTAN
SMILES CCCc1nc2c(C)cc(cc2n1Cc1ccc(cc1)-c1ccccc1C(O)=O)-c1nc2ccccc2n1C
InChI Key InChIKey=RMMXLENWKUUMAY-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 58 hits for monomerid = 50043280
Found 58 hits for monomerid = 50043280
Affinity DataKi: 0.230nMAssay Description:Displacement of [125I]-SI-Ang-2 from AT1 receptor in Rattus norvegicus Sprague-Dawley (rat) liver membranes after 2 hrMore data for this Ligand-Target Pair
Affinity DataKi: 0.230nMAssay Description:Displacement of [125I]SI-Ang2 from AT1 receptor in Sprague-Dawley rat liver membraneMore data for this Ligand-Target Pair
Affinity DataIC50: 0.330nMAssay Description:Displacement of [125I]Sar1 Ile8-Ang 2 from angiotensin 2 AT2 receptor after 180 mins by gamma countingMore data for this Ligand-Target Pair
Affinity DataIC50: 0.490nMAssay Description:Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor afterMore data for this Ligand-Target Pair
Affinity DataIC50: 0.490nMAssay Description:Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Displacement of [125I]-Sar1Ile8-angiotensin 2 from angiotensin 2 AT1 receptor (unknown origin) after 180 mins by gamma counting analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Displacement of [125I]Sar1 Ile8-Ang 2 from angiotensin 2 AT1 receptor after 180 mins by gamma countingMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Displacement of [125I]Sar1Ile8-Ang2 from angiotensin AT1 receptor after 180 mins by gamma countingMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of AT1 receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of AT1 receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of specific binding of [125I]angiotensin-II to angiotensin 1 receptor in rat lung membrane preparationMore data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Non-competitive inhibition of human recombinant CYP2J2 assessed as reduction in astemizole O-demethylation by LC-MS/MS method and Dixon plotMore data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Inhibition of angiotensin AT1 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 190nMAssay Description:Mixed type inhibition of human recombinant CYP2J2 assessed as reduction in astemizole O-demethylation by LC-MS/MS methodMore data for this Ligand-Target Pair
Affinity DataIC50: 420nMAssay Description:Inhibition of human recombinant CYP2J2 assessed as reduction in astemizole O-demethylation by LC-MS/MS methodMore data for this Ligand-Target Pair
Affinity DataIC50: 540nMAssay Description:Inhibition of human recombinant CYP2J2 assessed as reduction in astemizole O-demethylation reduction in astemizole O-demethylation after 30 mins by L...More data for this Ligand-Target Pair
Affinity DataIC50: 540nMAssay Description:Inhibition of human recombinant CYP2J2 assessed as reduction in astemizole O-demethylation after 30 mins by LC-MS/MS method in presence of 1 mM NADPHMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 700nMAssay Description:Displacement of Fluoromone from GST-tagged recombinant human PPARgamma ligand binding domain by LanthaScreen TR-FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 960nMAssay Description:Ki values for sodium fluorescein (10 uM) uptake in OATP1B3-transfected CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.03E+3nMAssay Description:Ki values for sodium fluorescein (10 uM) uptake in OATP1B1-transfected CHO cellsMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataKi: 1.08E+3nMAssay Description:Partial agonist activity at PPARgamma (unknown origin) by TR-FRET-based competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.26E+3nMAssay Description:pIC50 values for sodium fluorescein (10 uM) uptake in OATP1B3-transfected CHO cellsMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 1.52E+3nMAssay Description:Partial agonist activity at human PPARgamma-LBD/Gal4 DNA binding domain by transactivation assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 1.52E+3nMAssay Description:Partial agonist activity at human PPARgamma-LBD/Gal4 DNA binding domain by transactivation assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.86E+3nMAssay Description:pIC50 values for sodium fluorescein (10 uM) uptake in OATP1B1-transfected CHO cellsMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 2.02E+3nMAssay Description:Transactivation of GAL4-tagged human PPARgamma expressed in African green monkey COS7 cells after 36 hrs by luciferase reporter gene assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 2.02E+3nMAssay Description:Agonist activity at human PPARgamma expressed in african green monkey CV1 cells by Gal4 transactivation assayMore data for this Ligand-Target Pair
Affinity DataEC50: 3.20E+3nMAssay Description:Potentiation of human GlyR-alpha1 expressed in Xenopus laevis oocytes assessed as induction of glycine-activated currents after 1 to 4 days by two-el...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 3.40E+3nMAssay Description:Activation of Gal4-tagged human PPARgamma expressed in CHO cells by luciferase reporter gene assayMore data for this Ligand-Target Pair
Affinity DataEC50: 4.06E+3nMAssay Description:Agonist activity at mouse PPARgamma expressed in african green monkey CV1 cells by Gal4 transactivation assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 4.28E+3nMAssay Description:Transactivation of Gal4-fused human PPARgamma transfected in COS7 cells co-transfected with pGAL5-TK-pGL3 and pRennilla-CMV incubated for 39 hrs by d...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 4.30E+3nMAssay Description:Agonist activity at human PPARgammaDEF receptor expressed in african green monkey COS7 cells transfected with pGal5-TK-pGL3/pRenilla-CMV assessed as ...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 4.50E+3nMAssay Description:Activity at full length human PPARgamma transfected in CV1 cells assessed as transactivation activity after 24 hrs by PPRE-TK- luciferase reporter ge...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 4.70E+3nMAssay Description:Transactivation of GAL4-fused human PPARgamma ligand binding domain expressed in African green monkey COS7 cells after 42 hrs by dual luciferase repo...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 4.71E+3nMAssay Description:Transactivation at Gal4 fused PPARgamma LBD (unknown origin) expressed in African green monkey COS7 cells after 42 hrs by luciferase assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.78E+3nMAssay Description:Inhibition of CYP2C9 in human liver microsomes using tolbutamide substrate by LC-MS/MS methodMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataEC50: 4.97E+3nMAssay Description:Transactivation of Gal4 fused human PPARgamma LBD expressed in African green monkey COS7 cells after 42 hrs by dual luciferase reporter gene assayMore data for this Ligand-Target Pair
Affinity DataKi: 6.00E+3nMAssay Description:Inhibition of ACE (unknown origin) assessed as 3-Hydroxybutyril-glycil-glycil-glycine conversion to 3-hydroxybutyric acid after 60 mins by WST assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Displacement of radiolabeled GW-2433 from human PPARdelta ligand binding domain expressed in Escherichia coliMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Displacement of radiolabeled GW-2433 from human PPARalpha ligand binding domain expressed in Escherichia coliMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Agonist activity at mouse PPARdelta expressed in african green monkey CV1 cells by Gal4 transactivation assayMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Agonist activity at human PPARalpha expressed in african green monkey CV1 cells by Gal4 transactivation assayMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Agonist activity at human PPARdelta expressed in african green monkey CV1 cells by Gal4 transactivation assayMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Agonist activity at mouse PPARalpha expressed in african green monkey CV1 cells by Gal4 transactivation assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
University of Innsbruck
Curated by ChEMBL
University of Innsbruck
Curated by ChEMBL
Affinity DataIC50: 1.22E+4nMAssay Description:Displacement of radiolabeled ((5-{4-[Methyl-pyridin-2yl-amino)-ethoxy]-benzyl}-thiazolidine-2,4-dione) from human PPARgamma ligand binding domain exp...More data for this Ligand-Target Pair
Affinity DataIC50: 1.62E+4nMAssay Description:Inhibition of human BSEP expressed in fall armyworm sf9 cell plasma membrane vesicles assessed as reduction in vesicle-associated [3H]-taurocholate t...More data for this Ligand-Target Pair
Affinity DataIC50: 1.62E+4nMAssay Description:Inhibition of human BSEP overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-taurocholate in presence of ATP measured after 15 to ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.62E+4nMAssay Description:Inhibition of recombinant human BSEP expressed in baculovirus infected sf9 cell plasma membrane vesicles assessed as reduction in ATP-dependent [3H]-...More data for this Ligand-Target Pair
TargetBroad substrate specificity ATP-binding cassette transporter ABCG2(Human)
Federal University of Parana
Curated by ChEMBL
Federal University of Parana
Curated by ChEMBL
Affinity DataIC50: 1.69E+4nMAssay Description:Inhibition of human ABCG2 expressed in dog MDCK-II-BCRP cells mediated pheophorbide A efflux and measured after 60 mins using pheophorbide A as fluor...More data for this Ligand-Target Pair
Affinity DataIC50: 1.79E+4nMAssay Description:Inhibition of human MATE1-mediated ASP+ uptake expressed in HEK293 cells after 1.5 mins by fluorescence assayMore data for this Ligand-Target Pair