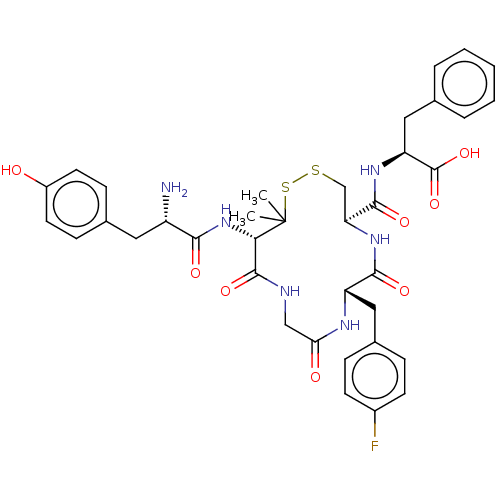
BDBM50043725 (S)-2-{[(4R,7S)-13-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-7-(4-fluoro-benzyl)-14,14-dimethyl-6,9,12-trioxo-1,2-dithia-5,8,11-triaza-cyclotetradecane-4-carbonyl]-amino}-3-phenyl-propionic acid::CHEMBL2372327
SMILES CC1(C)SSC[C@H](NC(=O)[C@H](Cc2ccc(F)cc2)NC(=O)CNC(=O)[C@@H]1NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(O)=O
InChI Key InChIKey=ZZJQYPCWCUPXMY-OLAIZJASSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50043725
Found 4 hits for monomerid = 50043725
Affinity DataIC50: 32nMAssay Description:Inhibitory concentration against electrically evoked contractions of guinea pig ileum longitudinal muscle-myenteric plexusMore data for this Ligand-Target Pair
Affinity DataIC50: 0.710nMAssay Description:Inhibitory concentration against delta opioid receptor of electrically induced smooth muscle contraction of mouse vas deferensMore data for this Ligand-Target Pair
Affinity DataIC50: 0.710nMAssay Description:Binding affinity towards delta opioid receptor was determined in rat brain using [H]-[p-Cl-Phe4]-DPDPE as radioligandMore data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Binding affinity towards mu opioid receptor was determined in rat brain using [3H]CTOP as radioligandMore data for this Ligand-Target Pair