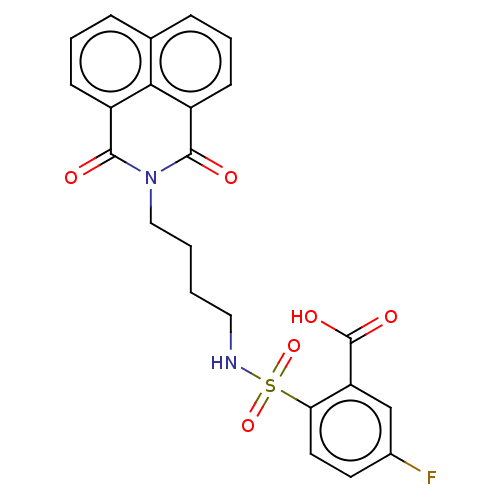
BDBM50056343 CHEMBL3322513
SMILES OC(=O)c1cc(F)ccc1S(=O)(=O)NCCCCN1C(=O)c2cccc3cccc(C1=O)c23
InChI Key InChIKey=OLNHPZQHEJGYNC-UHFFFAOYSA-N
Data 1 EC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 1 hit for monomerid = 50056343
Found 1 hit for monomerid = 50056343
TargetLysophosphatidic acid receptor 2(Human)
The University of Tennessee Health Science Center
Curated by ChEMBL
The University of Tennessee Health Science Center
Curated by ChEMBL
Affinity DataEC50: 0.150nMAssay Description:Agonist activity at human LPA2 expressed in LPA1xLPA2 double knockout mouse MEF cells by Fura-2AM dye based Ca2+ mobilization assayMore data for this Ligand-Target Pair