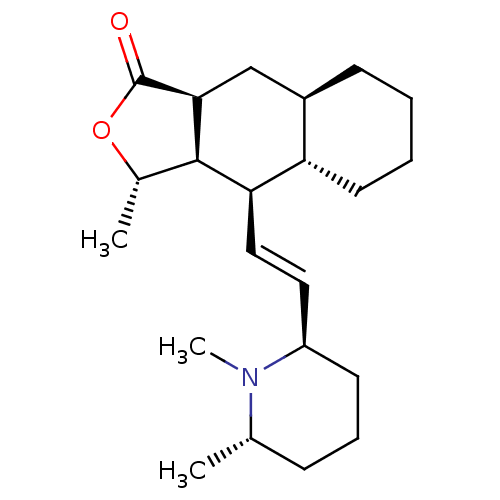
BDBM50076089 (3S,3aR,4R,4aS,8aR,9aS)-3-Methyl-4-[2-((R)-1-methyl-6-(S)-methyl-piperidin-2-yl)-vinyl]-decahydro-naphtho[2,3-c]furan-1-one::(3S,3aR,4R,4aS,8aR,9aS)-4-[(E)-2-((1S,2R,6S)-1,6-Dimethyl-piperidin-2-yl)-vinyl]-3-methyl-decahydro-naphtho[2,3-c]furan-1-one::(3S,3aR,4R,4aS,8aR,9aS)-4-[(E)-2-((2R,6S)-1,6-Dimethyl-piperidin-2-yl)-vinyl]-3-methyl-decahydro-naphtho[2,3-c]furan-1-one::(3S,3aR,4R,4aS,8aR,9aS)-4-[2-((3S,5R)-1,6-Dimethyl-piperidin-2-yl)-vinyl]-3-methyl-decahydro-naphtho[2,3-c]furan-1-one::4-[2-(1,6-Dimethyl-piperidin-2-yl)-vinyl]-3-methyl-decahydro-naphtho[2,3-c]furan-1-one::4-[2-[1,6-dimethyl-(2R,6S)-hexahydro-2-pyridinyl]-(E)-1-ethenyl]-3-methyl-(3S,3aR,4R,4aS,8aR,9aS)-perhydrobenzo[f]isobenzofuran-1-one::CHEMBL277642
SMILES C[C@@H]1OC(=O)[C@H]2C[C@H]3CCCC[C@@H]3[C@@H](\C=C\[C@H]3CCC[C@H](C)N3C)[C@@H]12
InChI Key InChIKey=FMPNFDSPHNUFOS-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 36 hits for monomerid = 50076089
Found 36 hits for monomerid = 50076089
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL