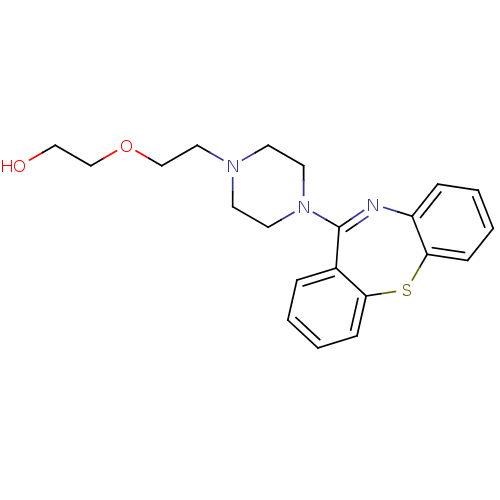
BDBM50095890 2-[2-(4-Dibenzo[b,f][1,4]thiazepin-11-yl-1-piperazinyl)ethoxy]ethanol::2-[2-(4-dibenzo[b,f][1,4]thiazepin-11-ylpiperazin-1-yl)ethoxy]ethanol::CHEMBL716::QUETIAPINE
SMILES OCCOCCN1CCN(CC1)C1=Nc2ccccc2Sc2ccccc12
InChI Key InChIKey=URKOMYMAXPYINW-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 218 hits for monomerid = 50095890
Found 218 hits for monomerid = 50095890
TargetAlpha-1A/Alpha-1B/Alpha-1D adrenergic receptor(Human)
Shanghai Institute of Pharmaceutical Industry
Curated by ChEMBL
Shanghai Institute of Pharmaceutical Industry
Curated by ChEMBL
Affinity DataKi: 7nMAssay Description:Binding affinity to adrenergic alpha1 receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 8.70nMAssay Description:Binding affinity to human cloned histamine H1 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 10nMAssay Description:Binding assay using 5-HT2A, Dopamine D2, SERT, αA1, 5-HT2C and H1 Receptors.More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Binding affinity to H1 histamine receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Displacement of [3H]prazosin from adrenergic alpha1 receptor in rat cerebral cortexMore data for this Ligand-Target Pair
Affinity DataKi: 19nMAssay Description:Binding affinity towards human histamine H1 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 20nMAssay Description:Binding assay using 5-HT2A, Dopamine D2, SERT, αA1, 5-HT2C and H1 Receptors.More data for this Ligand-Target Pair
Affinity DataKi: 21nMAssay Description:Binding affinity towards human H1 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 31nMAssay Description:Displacement of [3H]Ketanserin from 5-HT2A receptor in human brain tissue incubated for 60 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 40nMAssay Description:Displacement of [3H]pyrilamine from histaminergic H1 receptor guinea pig cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:Displacement of [3H]mepyramine from H1R in rat brainMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M1(Human)
Merck Sharp and Dohme Research Laboratories
Curated by ChEMBL
Merck Sharp and Dohme Research Laboratories
Curated by ChEMBL
Affinity DataKi: 56nMAssay Description:Binding affinity towards human M1 receptor.More data for this Ligand-Target Pair
TargetAlpha-1A/Alpha-1B/Alpha-1D adrenergic receptor(Human)
Shanghai Institute of Pharmaceutical Industry
Curated by ChEMBL
Shanghai Institute of Pharmaceutical Industry
Curated by ChEMBL
Affinity DataKi: 58nMAssay Description:Binding affinity towards human alpha-1 adrenergic receptorMore data for this Ligand-Target Pair
Affinity DataKi: 63nMAssay Description:Displacement of [3H]ketanserin from human 5HT2A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 63nMAssay Description:In vitro binding affinity for Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataKi: 63nMAssay Description:Displacement of [3H]LSD from human 5HT7 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 69nMAssay Description:Binding affinity towards human D2 dopamine receptor.More data for this Ligand-Target Pair
Affinity DataKi: 79nMAssay Description:Displacement of [3H]8-OH-DPAT from human 5HT1A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 82nMAssay Description:Binding affinity for human 5-hydroxytryptamine 2A receptorMore data for this Ligand-Target Pair
Affinity DataKi: 86nMAssay Description:Displacement of [3H]-LSD from human 5-HT2B receptor assessed as inhibition constantMore data for this Ligand-Target Pair
Affinity DataKi: 87nMAssay Description:Binding affinity towards human alpha-2 adrenergic receptorMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M1(Human)
Merck Sharp and Dohme Research Laboratories
Curated by ChEMBL
Merck Sharp and Dohme Research Laboratories
Curated by ChEMBL
Affinity DataKi: 100nMAssay Description:Binding affinity to human cloned muscarinic M1 receptorMore data for this Ligand-Target Pair