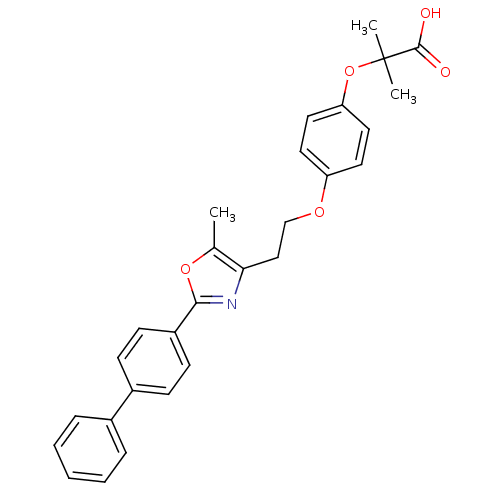
BDBM50100442 2-{4-[2-(2-Biphenyl-4-yl-5-methyl-oxazol-4-yl)-ethoxy]-phenoxy}-2-methyl-propionic acid::CHEMBL25259
SMILES Cc1oc(nc1CCOc1ccc(OC(C)(C)C(O)=O)cc1)-c1ccc(cc1)-c1ccccc1
InChI Key InChIKey=JDJHTJNBMZSSLK-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 10 hits for monomerid = 50100442
Found 10 hits for monomerid = 50100442
Affinity DataEC50: 150nMAssay Description:Binding affinity at human PPAR alphaMore data for this Ligand-Target Pair
Affinity DataEC50: 150nMAssay Description:Agonistic activity for Peroxisome proliferator activated receptor alphaMore data for this Ligand-Target Pair
Affinity DataEC50: 150nMAssay Description:Effective concentration against human peroxisome proliferator activated receptor alpha in Gal4 transactivation assayMore data for this Ligand-Target Pair
Affinity DataKi: 170nMAssay Description:Binding affinity for human peroxisome proliferator activated receptor alphaMore data for this Ligand-Target Pair
Affinity DataIC50: 174nMAssay Description:Binding affinity towards human peroxidase proliferator activated receptor alpha (hPPARalpha)More data for this Ligand-Target Pair
Affinity DataIC50: 548nMAssay Description:Binding affinity at human peroxidase proliferator activated receptor gamma (hPPARgamma)More data for this Ligand-Target Pair
Affinity DataKi: 550nMAssay Description:Binding affinity for human peroxisome proliferator activated receptor gammaMore data for this Ligand-Target Pair
Affinity DataEC50: 880nMAssay Description:Effective concentration against human peroxisome proliferator activated receptor gamma in Gal4 transactivation assayMore data for this Ligand-Target Pair
Affinity DataEC50: 880nMAssay Description:Agonistic activity for human Peroxisome proliferator activated receptor gammaMore data for this Ligand-Target Pair
Affinity DataEC50: 882nMAssay Description:Binding affinity at human PPAR gammaMore data for this Ligand-Target Pair