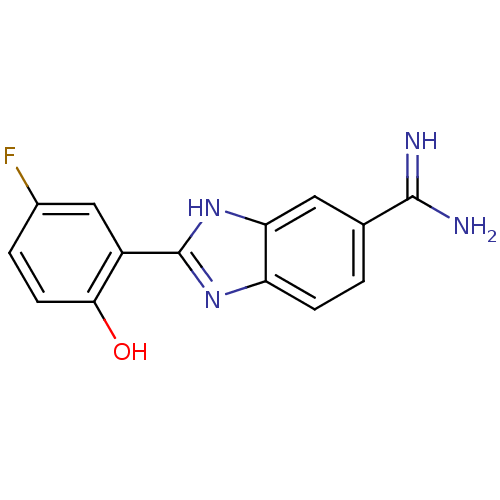
BDBM50102777 2-(5-Fluoro-2-hydroxy-phenyl)-1H-benzoimidazole-5-carboxamidine::2-{5-[AMINO(IMINIO)METHYL]-1H-BENZIMIDAZOL-2-YL}-4-FLUOROBENZENOLATE::CHEMBL92277
SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(F)ccc1O
InChI Key InChIKey=JOGOACSSLJEREC-UHFFFAOYSA-N
Data 6 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50102777
Found 6 hits for monomerid = 50102777
Affinity DataKi: 2.80E+3nMAssay Description:ComInhibition of Human Serine Protease Urokinase Plasminogen Activator (u-PA).More data for this Ligand-Target Pair
Affinity DataKi: 6.80E+3nMAssay Description:Binding affinity against human coagulation factor XMore data for this Ligand-Target Pair
Affinity DataKi: 1.00E+4nMAssay Description:Inhibition of Human Serine Protease Trypsin.More data for this Ligand-Target Pair
Affinity DataKi: 2.10E+4nMAssay Description:Inhibition of Human Serine Protease Plasmin.More data for this Ligand-Target Pair
Affinity DataKi: 3.30E+4nMAssay Description:Inhibition of Human Serine Protease Thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 4.60E+4nMAssay Description:Inhibition of Human Serine Protease tissue type Plasminogen Activator (t-PA).More data for this Ligand-Target Pair