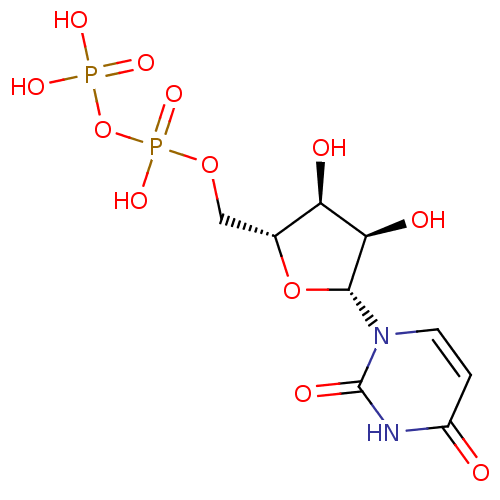
BDBM50118239 CHEMBL130266::UDP::Uridine diphosphate::uridine 5'-(trihydrogen diphosphate)
SMILES O[C@@H]1[C@@H](COP(O)(=O)OP(O)(O)=O)O[C@H]([C@@H]1O)n1ccc(=O)[nH]c1=O
InChI Key InChIKey=XCCTYIAWTASOJW-XVFCMESISA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 23 hits for monomerid = 50118239
Found 23 hits for monomerid = 50118239
TargetRibonuclease pancreatic(Bison bison (American bison))
National Hellenic Research Foundation
Curated by ChEMBL
National Hellenic Research Foundation
Curated by ChEMBL
Affinity DataKi: 6.47E+5nMpH: 6.0Assay Description:Inhibition of bovine pancreatic ribonuclease A assessed as enzyme activity by spectrophotometric method pH 6More data for this Ligand-Target Pair
Affinity DataKi: 1.05E+6nMpH: 6.0Assay Description:Inhibition of bovine seminal ribonuclease assessed as enzyme activity by spectrophotometric method at pH 6More data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 300nMAssay Description:Agonist activity at human recombinant P2Y6 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate production by scintillation pr...More data for this Ligand-Target Pair
TargetP2Y purinoceptor 14(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 160nMAssay Description:Agonist activity at human recombinant P2Y14 receptor expressed in HEK293 cells assessed as [3H]inositol phosphate production by scintillation proximi...More data for this Ligand-Target Pair
TargetUDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit(Homo sapiens (Human))
University Of British Columbia
Curated by ChEMBL
University Of British Columbia
Curated by ChEMBL
Affinity DataIC50: 1.80E+3nMAssay Description:Inhibition of human OGTMore data for this Ligand-Target Pair
Affinity DataEC50: 1.40E+4nMAssay Description:Agonist activity at human recombinant P2Y4 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate intracellular accumulation by ...More data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 48nMAssay Description:Agonist activity at human recombinant P2Y6 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate intracellular accumulation by ...More data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Agonist activity at rat P2Y purinoceptor 6 expressed in human 1321N1 cells assessed as increase in intracellular calcium concentration by spectrofluo...More data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 300nMAssay Description:The compound was evaluated for agonist activity against phospholipase C coupled recombinant human P2Y purinoceptor 6 (P2Y6)More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+5nMAssay Description:Evaluated for agonist activity against phospholipase C coupled P2Y purinoceptor 1 (P2Y1) of human erythrocytesMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+5nMAssay Description:Evaluated for agonist activity against phospholipase C coupled recombinant human P2Y purinoceptor 2 (P2Y2)More data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 42nMAssay Description:Agonist activity at human P2Y6 receptor expressed in 1321N1 cells assessed as IP accumulation by SPAMore data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 13nMAssay Description:Agonist activity at human recombinant P2Y6 receptor expressed in 1321N1 cells assessed as PLC-mediated [3H]IP productionMore data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 140nMAssay Description:Agonist activity at human P2Y6 receptor expressed in human 1321N1 cells assessed as calcium elevation by fura2/AM assayMore data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Agonist activity at rat P2Y6 receptor expressed in human 1321N1 cells assessed as increase in intracellular calcium concentration by dual-excitation ...More data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 300nMAssay Description:Agonist activity at human P2Y6 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate accumulation by scintillation proximity as...More data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 530nMAssay Description:Agonist activity at human P2Y6 receptor expressed in human 1321N1 cells coexpressing phospholipase C-activating Gq protein assessed as [3H]inositol p...More data for this Ligand-Target Pair
TargetP2Y purinoceptor 14(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 160nMAssay Description:Agonist activity at human P2Y14 receptor expressed in HEK293 cells coexpressing phospholipase C-activating Gi protein cells assessed as inhibition of...More data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 42nMAssay Description:Agonist activity at human P2Y6R expressed in 1321N1 cells assessed as calcium mobilization measured by microplate reader methodMore data for this Ligand-Target Pair
TargetUDP-galactopyranose mutase(Klebsiella pneumoniae)
Guizhou University Of Traditional Chinese Medicine
Curated by ChEMBL
Guizhou University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataKd: 5.40E+4nMAssay Description:Inhibition of Klebsiella pneumoniae UGM expressed in Escherichia coli BL21(DE3) assessed as dissociation constant by fluorescence polarization assayMore data for this Ligand-Target Pair
TargetUDP-galactopyranose mutase(Mycobacterium tuberculosis H37Rv)
Guizhou University Of Traditional Chinese Medicine
Curated by ChEMBL
Guizhou University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataKd: 3.50E+4nMAssay Description:Inhibition of Mycobacterium tuberculosis UGM expressed in Escherichia coli BL21(DE3) assessed as dissociation constant by fluorescence polarization a...More data for this Ligand-Target Pair
Affinity DataEC50: 1.65E+4nMAssay Description:Agonist activity at human recombinant P2Y2 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate intracellular accumulation by ...More data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 86nMAssay Description:Stimulation of phospholipase C in 1321N1 astrocytoma cells transfected with human P2Y6 receptorMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)