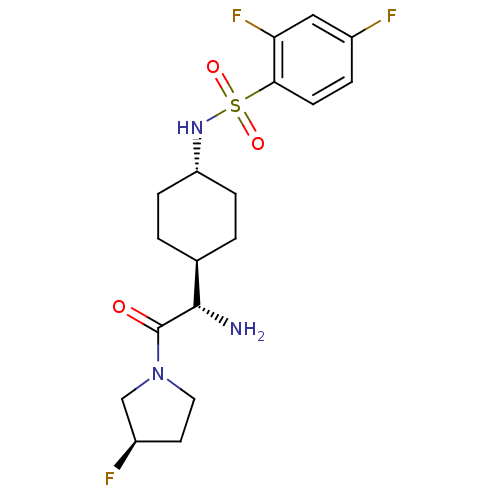
BDBM50140522 CHEMBL283309::N-{4-[(S)-1-Amino-2-((R)-3-fluoro-pyrrolidin-1-yl)-2-oxo-ethyl]-cyclohexyl}-2,4-difluoro-benzenesulfonamide
SMILES N[C@@H]([C@H]1CC[C@@H](CC1)NS(=O)(=O)c1ccc(F)cc1F)C(=O)N1CC[C@@H](F)C1
InChI Key InChIKey=QTZTUCOZZMQUFY-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50140522
Found 3 hits for monomerid = 50140522
Affinity DataIC50: 58nMAssay Description:Inhibitory activity against human Dipeptidylpeptidase IVMore data for this Ligand-Target Pair
Affinity DataIC50: 9.40E+3nMAssay Description:Inhibitory activity against human quiescent cell proline dipeptidase (QPP) enzymeMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 5.20E+4nMAssay Description:Binding affinity towards human ERG potassium ion channel was determinedMore data for this Ligand-Target Pair