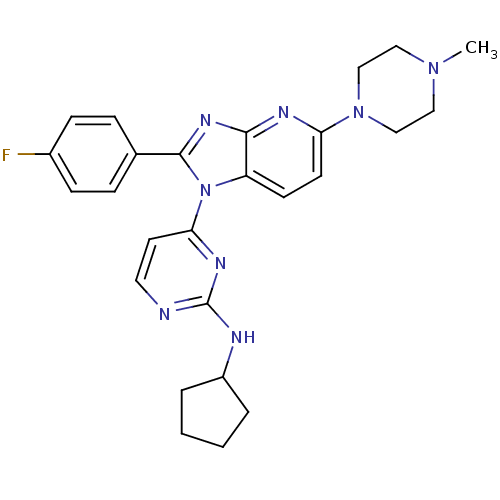
BDBM50148692 CHEMBL120185::Cyclopentyl-{4-[2-(4-fluoro-phenyl)-5-(4-methyl-piperazin-1-yl)-imidazo[4,5-b]pyridin-1-yl]-pyrimidin-2-yl}-amine
SMILES CN1CCN(CC1)c1ccc2n(c(nc2n1)-c1ccc(F)cc1)-c1ccnc(NC2CCCC2)n1
InChI Key InChIKey=BARFMAOTOPEJFY-UHFFFAOYSA-N
Data 19 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 19 hits for monomerid = 50148692
Found 19 hits for monomerid = 50148692
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human Epidermal growth factor receptor, HER-1More data for this Ligand-Target Pair
TargetTyrosine-protein kinase ABL1(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human c-AblMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >8.00E+4nMAssay Description:Inhibition of human prostaglandin G/H synthase 1, COX-1More data for this Ligand-Target Pair
TargetProto-oncogene tyrosine-protein kinase Src(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human c-SrcMore data for this Ligand-Target Pair
TargetCytochrome P450 2D6(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >2.00E+3nMAssay Description:Inhibition of human cytochrome P450 2D6More data for this Ligand-Target Pair
TargetMitogen-activated protein kinase 14(Mus musculus (mouse))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 15nMAssay Description:Inhibition of murine phosphorylated His-Mitogen-activated protein kinase p38 alpha.More data for this Ligand-Target Pair
TargetInsulin-like growth factor 1 receptor(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human insulin-like growth factor I receptorMore data for this Ligand-Target Pair
TargetRAF proto-oncogene serine/threonine-protein kinase(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human RAF proto-oncogene serine/threonine-protein kinaseMore data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human vascular endothelial growth factor receptor 2More data for this Ligand-Target Pair
TargetMitogen-activated protein kinase 9(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 47nMAssay Description:Inhibition of human c-Jun N-terminal kinase 2More data for this Ligand-Target Pair
TargetMast/stem cell growth factor receptor Kit(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human c-Kit kinaseMore data for this Ligand-Target Pair
TargetCytochrome P450 2C9(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >2.00E+3nMAssay Description:Inhibition of human cytochrome P450 2C9More data for this Ligand-Target Pair
TargetCytochrome P450 1A2(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >2.00E+3nMAssay Description:Inhibition of human cytochrome P450 1A2More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 8.70E+3nMAssay Description:Inhibition of human epidermal growth factor receptorMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >2.00E+3nMAssay Description:Inhibition of human cytochrome P450 3A4More data for this Ligand-Target Pair
TargetCyclin-dependent kinase 1(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human cyclin-dependent kinase 1More data for this Ligand-Target Pair
TargetMitogen-activated protein kinase 8(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 1.50E+3nMAssay Description:Inhibition of human c-Jun N-terminal kinase 1More data for this Ligand-Target Pair
TargetHepatocyte growth factor receptor(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human Met proto-oncogene tyrosine kinaseMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor/Receptor tyrosine-protein kinase erbB-2(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human HER-2More data for this Ligand-Target Pair