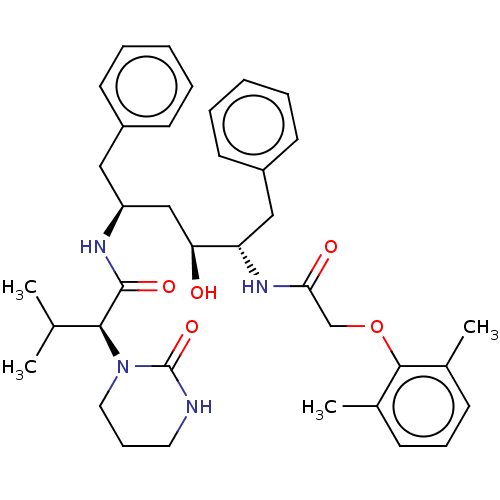
BDBM50180655 A-157378-0::A-157378.0::ABT-378::CHEBI:31781::Kaletra::Lopinavir::med.21724, Compound 185
SMILES CC(C)[C@H](N1CCCNC1=O)C(=O)N[C@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)COc1c(C)cccc1C)Cc1ccccc1
InChI Key InChIKey=KJHKTHWMRKYKJE-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 36 hits for monomerid = 50180655
Found 36 hits for monomerid = 50180655
Affinity DataKd: 0.00130nMAssay Description:Binding affinity to HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.00500nMAssay Description:Inhibition of wild type HIV1 protease by FRETMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
University of Massachusetts Medical School
Curated by ChEMBL
University of Massachusetts Medical School
Curated by ChEMBL
Affinity DataKi: <0.00500nMAssay Description:Inhibition of HIV1 NL4-3 protease expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing protease cleavage ...More data for this Ligand-Target Pair
Affinity DataKi: 0.00500nMAssay Description:Binding affinity to wild type HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.0180nMAssay Description:Inhibition of wild-type HIV1 BH10 protease expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.0260nMAssay Description:Inhibition of HIV1 recombinant protease D30N/N88D mutant expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.0290nMAssay Description:Inhibition of HIV1 recombinant protease M46I/A71V/V82T/I84V mutant expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.0510nMAssay Description:Inhibition of HIV1 NL4-3 protease I84V mutant expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing protea...More data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.0600nMAssay Description:Inhibition of HIV1 recombinant protease A71V/V82T/I84V mutant expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.0610nMAssay Description:Inhibition of HIV1 NL4-3 protease I50V/A71V mutant expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing p...More data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:Inhibition of HIV1 recombinant protease L10F/L19I/K20R/L33F/E35D/M36I/R41K/F53L/I54V/L63P/H69K/A71V/T74P/I84V/L89M/L90M/I93L mutant expressed in Esch...More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Inhibition of HIV1 protease expressed in Escherichia coli by fluorometric assayMore data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 2.40nMAssay Description:Inhibition of HIV1 recombinant protease V32I/I47A mutant expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Inhibition of HIV-1 NL4-3 wild type protease expressed in Escherichia coli preincubated for 20 mins by FRET analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Inhibition of HIV1 subtype C protease expressed in Escherichia coli BL21 assessed as hydrolysis of substrate using chromogenic substrate Lys-Ala-Arg-...More data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataIC50: 25nMAssay Description:Inhibition of HIV1 subtype C protease Q7K mutant expressed in Escherichia coli BL21 (DE3) pLysS using Lys-Ala-Arg-Val-Nle-p-nitro-Phe-Glu-Ala-Nle-NH2...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Inhibition of South African HIV1 subtype C protease expressed in Escherichia coli BL21S4 (DE3)pLysS cells using Lys-Ala-Arg-Val-Nle-p-nitro-Phe-Glu-A...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Inhibition of wild type C-South African Human immunodeficiency virus 1 protease using chromogenic peptide H-1048 as substrate by UV spectrophotometri...More data for this Ligand-Target Pair
Affinity DataEC50: 1.16E+3nMAssay Description:Inhibition of protease L10F/V32I/M46I/I47V/Q58E/I84V mutant in HIV1 A17 infected in human MT4 cells assessed as reduction in virus-induced cytopathic...More data for this Ligand-Target Pair
Affinity DataEC50: 6.60E+3nMAssay Description:This is a review article.More data for this Ligand-Target Pair
TargetBroad substrate specificity ATP-binding cassette transporter ABCG2(Human)
Federal University of Parana
Curated by ChEMBL
Federal University of Parana
Curated by ChEMBL
Affinity DataIC50: 7.60E+3nMAssay Description:Inhibition of human ABCG2 expressed in dog MDCK-II-BCRP cells mediated pheophorbide A efflux preincubated with PhA followed by compound addition and ...More data for this Ligand-Target Pair
Affinity DataEC50: 8.00E+3nMAssay Description:This is a review article.More data for this Ligand-Target Pair
Affinity DataEC50: 8.00E+3nMAssay Description:This is a review article.More data for this Ligand-Target Pair
Affinity DataIC50: 9.12E+3nMAssay Description:Ten-point DRCs were generated for each drug. Vero cells were seeded at 1.2 × 104 cells per well in DMEM, supplemented with 2% FBS and 1× ...More data for this Ligand-Target Pair
Affinity DataIC50: 9.12E+3nMAssay Description:Inhibition of SARS-CoV-2 main proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 1.50E+4nMAssay Description:This is a review article. Please point to the original journal.More data for this Ligand-Target Pair
Affinity DataIC50: 1.53E+4nMAssay Description:Ten-point DRCs were generated for each drug. Vero cells were seeded at 1.2 × 104 cells per well in DMEM, supplemented with 2% FBS and 1× ...More data for this Ligand-Target Pair
Affinity DataEC50: 1.71E+4nMAssay Description:This is a review article.More data for this Ligand-Target Pair
Affinity DataIC50: 1.73E+4nMAssay Description:Inhibition of human BSEP expressed in fall armyworm sf9 cell plasma membrane vesicles assessed as reduction in vesicle-associated [3H]-taurocholate t...More data for this Ligand-Target Pair
Affinity DataIC50: 1.73E+4nMAssay Description:Inhibition of human BSEP overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-taurocholate in presence of ATP measured after 15 to ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of SARS-CoV-2 MProMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+4nMAssay Description:Inhibition of human MRP3 overexpressed in Sf9 insect cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ...More data for this Ligand-Target Pair
Affinity DataEC50: 2.44E+4nMAssay Description:This is a review article.More data for this Ligand-Target Pair
Affinity DataIC50: 4.70E+4nMAssay Description:Inhibition of human MRP4 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and...More data for this Ligand-Target Pair
Affinity DataIC50: 1.33E+5nMAssay Description:Inhibition of human MRP2 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and...More data for this Ligand-Target Pair