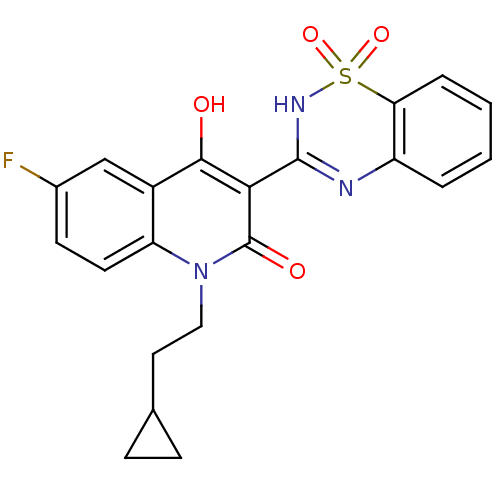
BDBM50181559 1-(2-CYCLOPROPYLETHYL)-3-(1,1-DIOXIDO-2H-1,2,4-BENZOTHIADIAZIN-3-YL)-6-FLUORO-4-HYDROXYQUINOLIN-2(1H)-ONE::1-(2-Cyclopropyl-ethyl)-3-(1,1-dioxo-1,4-dihydro-1lambda*6*-benzo[1,2,4]thiadiazin-3-yl)-6-fluoro-4-hydroxy-1H-quinolin-2-one::1-(2-cyclopropylethyl)-3-(1,1-dioxo-1,4-dihydrobenzo[1,2,4]-thiadiazin-3-yl) -6-fluoro-4-hydroxy-1-quinolin-2-one::CHEMBL372357
SMILES c1ccc2c(c1)N=C(NS2(=O)=O)C3=C(c4cc(ccc4N(C3=O)CCC5CC5)F)O
InChI Key InChIKey=QEMCDXCXSVPAAB-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 11 hits for monomerid = 50181559
Found 11 hits for monomerid = 50181559
TargetRNA-directed RNA polymerase(Hepatitis C virus genotype 1b (isolate Con1) (HCV))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibitory activity against HCV 1b NS5B RNA polymeraseMore data for this Ligand-Target Pair
TargetRNA-directed RNA polymerase(Hepatitis C virus genotype 1b (isolate Con1) (HCV))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibitory activity against HCV delta 21 NS5B RNA polymerase by SPA assayMore data for this Ligand-Target Pair
TargetRNA-directed RNA polymerase(Hepatitis C virus genotype 1b (isolate Con1) (HCV))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibitory activity against HCV 2a NS5B RNA polymeraseMore data for this Ligand-Target Pair
TargetRNA-directed RNA polymerase(Hepatitis C virus genotype 1b (isolate Con1) (HCV))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataEC50: 38nMAssay Description:Inhibition of HCV RNA replication in Huh7 cellsMore data for this Ligand-Target Pair
TargetRNA-directed RNA polymerase(Hepatitis C virus genotype 1b (isolate Con1) (HCV))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 49nMAssay Description:Inhibitory activity against HCV 1a NS5B RNA polymeraseMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of CYP2C9 isozymeMore data for this Ligand-Target Pair
Affinity DataEC50: 5.30E+3nMAssay Description:Effect on PXR activityMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibitory activity against human DNA-polymerase-alphaMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibitory activity against virus GBV-B polymeraseMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibitory activity against human DNA-polymerase-betaMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)