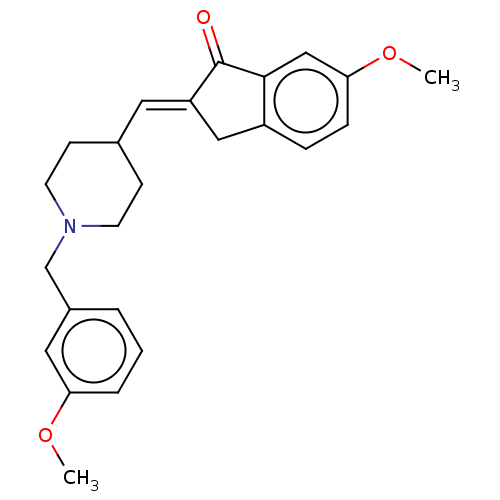
BDBM50182438 CHEMBL3819113
SMILES COc1ccc2C\C(=C/C3CCN(Cc4cccc(OC)c4)CC3)C(=O)c2c1
InChI Key InChIKey=VRHAQRBXZOTBMX-UDWIEESQSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50182438
Found 4 hits for monomerid = 50182438
Affinity DataKi: 3.89E+3nMAssay Description:Non-competitive inhibition of horse serum BuChE using butyrylthiocoline iodide as substrate incubated for 20 mins by Lineweaver-Burk plot analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.19E+5nMAssay Description:Non-competitive inhibition of human erythrocytes AchE using acetylthiocholine iodide as substrate incubated for 20 mins by Lineweaver-Burk plot analy...More data for this Ligand-Target Pair
Affinity DataIC50: 1.62E+3nMAssay Description:Inhibition of horse serum BuChE using butyrylthiocoline iodide as substrate incubated for 20 mins by Ellman methodMore data for this Ligand-Target Pair
Affinity DataIC50: 3.45E+4nMAssay Description:Inhibition of human erythrocytes AchE using acetylthiocholine iodide as substrate incubated for 20 mins by Ellman methodMore data for this Ligand-Target Pair