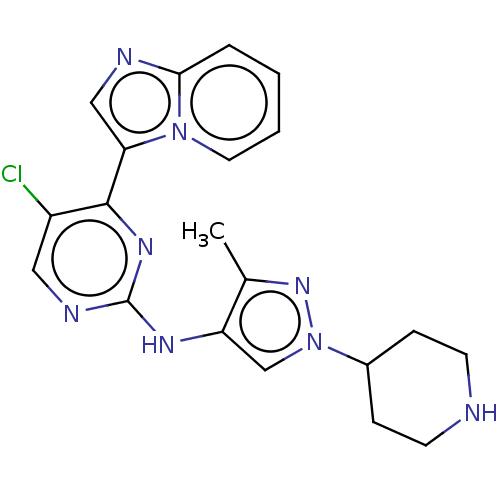
BDBM50184470 CHEMBL3823007
SMILES Cc1nn(cc1Nc1ncc(Cl)c(n1)-c1cnc2ccccn12)C1CCNCC1
InChI Key InChIKey=JVFLXTNKVBOVBS-UHFFFAOYSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50184470
Found 4 hits for monomerid = 50184470
Affinity DataIC50: 38nMAssay Description:Inhibition of recombinant human IGF-1R using fluorescent labeled FL-KKSRGDYMTMQIG-CONH2 as substrate after 1 hr 50 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 54nMAssay Description:Inhibition of IGF1-induced human IGF1R autophosphorylation expressed in IGF-1R knock-out mouse fibroblastsMore data for this Ligand-Target Pair
Affinity DataIC50: 392nMAssay Description:Inhibition of CDK2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+4nMAssay Description:Inhibition of human ERG expressed in CHOK1 cells after 6 mins by electrophysiology assayMore data for this Ligand-Target Pair