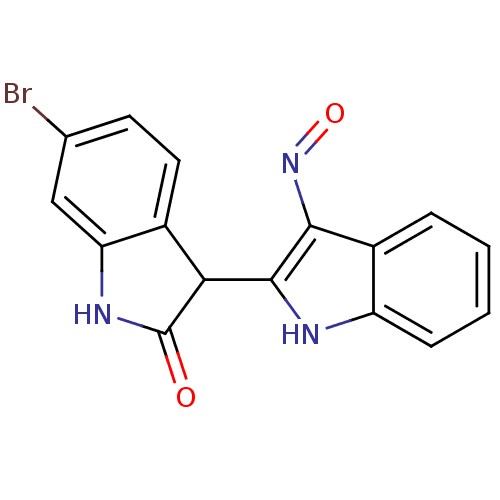
BDBM7401 (2 Z,3 E)-6-Bromoindirubin-3 -oxime::(3E)-6-bromo-3-[3-(hydroxyamino)indol-2-ylidene]oxindole::6-bromo-3-[(2Z,3E)-3-(hydroxyimino)-2,3-dihydro-1H-indol-2-ylidene]-2,3-dihydro-1H-indol-2-one::6-bromoindirubin-3 -oxime::6BIO::CHEMBL409450::indirubin deriv. 7a
SMILES Brc1ccc2C(C(=O)Nc2c1)c1[nH]c2ccccc2c1N=O
InChI Key InChIKey=OLOJSTRISMGUEK-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 25 hits for monomerid = 7401
Found 25 hits for monomerid = 7401
Affinity DataIC50: 5nMT: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of porcine brain GSK3beta using YRRAAVPPSPSLSRHSSPHQSpEDEEE as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Binding affinity to GSK3alphaMore data for this Ligand-Target Pair
Affinity DataIC50: 83nMAssay Description:Inhibition of mammalian CDK5/p25More data for this Ligand-Target Pair
Affinity DataIC50: 83nMAssay Description:Inhibition of human recombinant CDK5More data for this Ligand-Target Pair
Affinity DataIC50: 83nMT: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
Affinity DataIC50: 83nMAssay Description:inhibition of human recombinant CDK5/p25 using histone H1 as substrate by scintillation counting in presence of [gamma-33P]ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 1/G2/mitotic-specific cyclin-B1/G2/mitotic-specific cyclin-B2/G2/mitotic-specific cyclin-B3(Human)
Rockefeller University
Curated by ChEMBL
Rockefeller University
Curated by ChEMBL
Affinity DataIC50: 320nMAssay Description:Inhibition of CDK1/cyclinBMore data for this Ligand-Target Pair
Affinity DataIC50: 320nMAssay Description:Inhibition of CDK1/cyclin BMore data for this Ligand-Target Pair
Affinity DataIC50: 320nMpH: 7.2 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
TargetDual specificity tyrosine-phosphorylation-regulated kinase 1A(Rat)
Rajiv Gandhi Proudyogiki Vishwavidyalaya
Curated by ChEMBL
Rajiv Gandhi Proudyogiki Vishwavidyalaya
Curated by ChEMBL
Affinity DataIC50: 520nMAssay Description:Inhibition of rat recombinant GST-fused DYRK1A expressed in Escherichia coli using KKISGRLSPIMTEQ as substrate by scintillation counting in presence ...More data for this Ligand-Target Pair
TargetInterferon-induced, double-stranded RNA-activated protein kinase(Human)
Weill Cornell Medical College
Curated by ChEMBL
Weill Cornell Medical College
Curated by ChEMBL
Affinity DataIC50: 600nMAssay Description:Inhibition of human recombinant PKR autophosphorylation using poly[I:C] after 10 mins by luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Inhibition of porcine brain CK1 using RRKHAAIGpSAYSITA as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMpH: 7.5 T: 2°CAssay Description:PDK1 protein kinase was expressed in Sf9 insect cells as human recombinant GST-fusion protein by means of the baculovirus expression systems. Protei...More data for this Ligand-Target Pair
TargetDual specificity tyrosine-phosphorylation-regulated kinase 1A(Rat)
Rajiv Gandhi Proudyogiki Vishwavidyalaya
Curated by ChEMBL
Rajiv Gandhi Proudyogiki Vishwavidyalaya
Curated by ChEMBL
Affinity DataIC50: 1.70E+3nMAssay Description:Inhibition of rat recombinant GST-fused DYRK1A expressed in Escherichia coli using KKISGRLSPIMTEQ as substrate after 30 mins by scintillation countin...More data for this Ligand-Target Pair
TargetDual specificity tyrosine-phosphorylation-regulated kinase 2(Human)
University of Athens
Curated by ChEMBL
University of Athens
Curated by ChEMBL
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of human recombinant GST-fused DYRK2 expressed in Escherichia coli using KKISGRLSPIMTEQ as substrate after 30 mins by scintillation counti...More data for this Ligand-Target Pair
TargetDual specificity protein kinase CLK1(Human)
Rajiv Gandhi Proudyogiki Vishwavidyalaya
Curated by ChEMBL
Rajiv Gandhi Proudyogiki Vishwavidyalaya
Curated by ChEMBL
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of human recombinant GST-fused CLK1 expressed in Escherichia coli using GRSRSRSRSRSR as substrateMore data for this Ligand-Target Pair
TargetBeta-galactosidase(Escherichia coli)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 4.08E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, C...More data for this Ligand-Target Pair
Affinity DataEC50: 4.49E+4nMAssay Description:Keywords: Group A streptococcus, GAS, streptokinase, expression, virulence, inhibition, dose response, EC50 Assay Overview: The goal of this assa...More data for this Ligand-Target Pair
Affinity DataIC50: 6.43E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetC-C chemokine receptor type 6(Human)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 6.66E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair
TargetSentrin-specific protease 8(Human)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetSentrin-specific protease 6(Human)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair