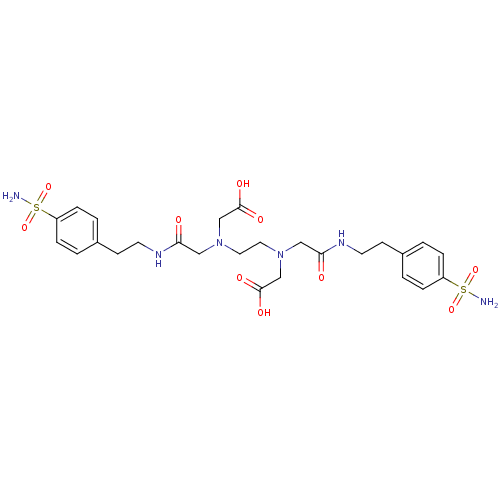
BDBM50228328 ([2-(Carboxymethyl-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-ethyl]-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-acetic acid; compound with Zn complex::([2-(carboxymethyl-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-ethyl]-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-acetic acid::CHEMBL1795054::CHEMBL34526
SMILES NS(=O)(=O)c1ccc(CCNC(=O)CN(CCN(CC(O)=O)CC(=O)NCCc2ccc(cc2)S(N)(=O)=O)CC(O)=O)cc1
InChI Key InChIKey=NIYUBSMBQZFXKI-UHFFFAOYSA-N
Data 10 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 10 hits for monomerid = 50228328
Found 10 hits for monomerid = 50228328
TargetCarbonic anhydrase 2(Human)
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Affinity DataKi: 0.770nMAssay Description:Inhibition of human recombinant CA2 by CO2 hydration stopped flow assayMore data for this Ligand-Target Pair
TargetCarbonic anhydrase 12(Human)
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Affinity DataKi: 2.80nMAssay Description:Inhibition of human recombinant CA12 by CO2 hydration stopped flow assayMore data for this Ligand-Target Pair
TargetCarbonic anhydrase 2(Human)
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Affinity DataKi: 8nMAssay Description:Inhibitory activity against Human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetCarbonic anhydrase 9(Human)
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Affinity DataKi: 8.20nMAssay Description:Inhibition of human recombinant CA9 by CO2 hydration stopped flow assayMore data for this Ligand-Target Pair
TargetCarbonic anhydrase 1(Human)
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Affinity DataKi: 9.40nMAssay Description:Inhibition of human recombinant CA1 by CO2 hydration stopped flow assayMore data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Inhibitory activity against bovine carbonic anhydrase IVMore data for this Ligand-Target Pair
TargetCarbonic anhydrase 2(Human)
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Affinity DataKi: 33nMAssay Description:Inhibitory activity against Human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetCarbonic anhydrase 1(Human)
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Affinity DataKi: 39nMAssay Description:Inhibitory activity against Human carbonic anhydrase IMore data for this Ligand-Target Pair
Affinity DataKi: 54nMAssay Description:Inhibitory activity against bovine carbonic anhydrase IVMore data for this Ligand-Target Pair
TargetCarbonic anhydrase 1(Human)
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Ecole Nationale Sup£Rieure De Chimie De Montpellier
Curated by ChEMBL
Affinity DataKi: 400nMAssay Description:Inhibitory activity against Human carbonic anhydrase IMore data for this Ligand-Target Pair