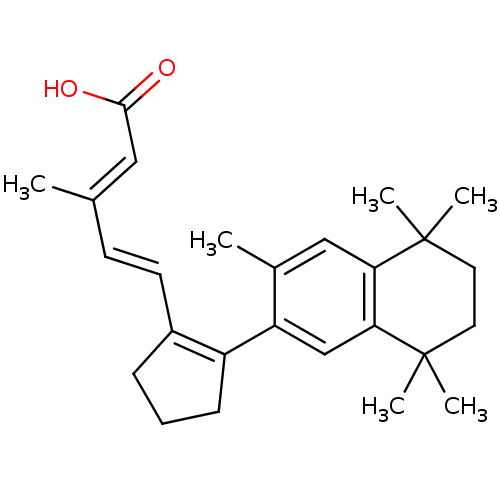
BDBM50290193 3-Methyl-5-[2-(3,5,5,8,8-pentamethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)-cyclopent-1-enyl]-penta-2,4-dienoic acid::CHEMBL312737
SMILES C\C(\C=C\C1=C(CCC1)c1cc2c(cc1C)C(C)(C)CCC2(C)C)=C/C(O)=O
InChI Key InChIKey=GMUNIMQISYVYQW-PKAMWHFSSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 9 hits for monomerid = 50290193
Found 9 hits for monomerid = 50290193
Affinity DataKi: 5nMAssay Description:Inhibition of [3H]-9-cis-RA binding to Retinoid X receptor RXR alphaMore data for this Ligand-Target Pair
Affinity DataKi: 22nMAssay Description:Inhibition of [3H]-9-cis-RA binding to Retinoid X receptor RXR betaMore data for this Ligand-Target Pair
Affinity DataKi: 25nMAssay Description:Inhibition of [3H]-9-cis-RA binding to Retinoid X receptor RXR betaMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-ATRA binding to retinoic acid receptor RAR alphaMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-ATRA binding to retinoic acid receptor RAR betaMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-ATRA binding to Retinoic acid receptor RAR gammaMore data for this Ligand-Target Pair
Affinity DataEC50: 95nMAssay Description:Inhibition of [3H]-9-cis-RA binding to Retinoid X receptor RXR gammaMore data for this Ligand-Target Pair
Affinity DataEC50: 191nMAssay Description:Inhibition of [3H]-ATRA binding to Retinoic acid receptor RAR gammaMore data for this Ligand-Target Pair
Affinity DataEC50: 128nMAssay Description:Effective potency in transcriptional activation assay in CV-1 cells expressing Retinoid X receptor RXR gammaMore data for this Ligand-Target Pair