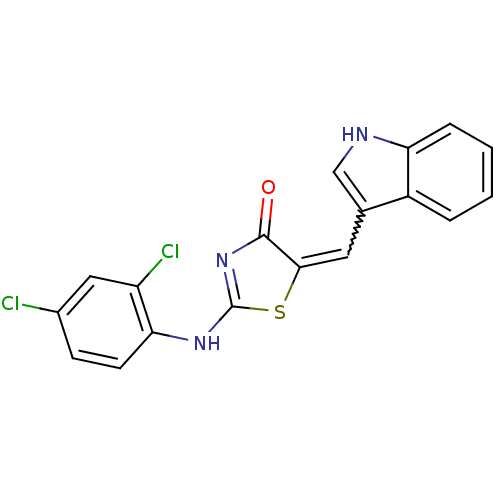
BDBM50296454 5-((1H-indol-3-yl)methylene)-2-(2,4-dichlorophenylamino)thiazol-4(5H)-one::CHEMBL549295::cid_5530131
SMILES Clc1ccc(NC2=NC(=O)C(S2)=Cc2c[nH]c3ccccc23)c(Cl)c1
InChI Key InChIKey=OYSYEYPBKTUHFY-UHFFFAOYSA-N
Data 6 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50296454
Found 6 hits for monomerid = 50296454
Affinity DataIC50: 1.50E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of N-terminal 6xHis-tagged (378-566) catalytic domain of Cdc25B expressed in Escherichia coli after 20 mins by by fluorescent plate reader...More data for this Ligand-Target Pair
Affinity DataIC50: 2.64E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
Affinity DataIC50: 1.26E+4nMAssay Description:Inhibition of N-terminal 6xHis-tagged human Cdc25A (336-523) catalytic domain expressed in Escherichia coli after 20 mins by fluorescent plate reader...More data for this Ligand-Target Pair
Target26S proteasome non-ATPase regulatory subunit 14(Human)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 3.65E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford Burnham Medical Research Institute (SBMRI, La Jolla, CA...More data for this Ligand-Target Pair
Affinity DataIC50: 3.83E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford Burnham Medical Research Institute (SBMRI, La Jolla, CA...More data for this Ligand-Target Pair