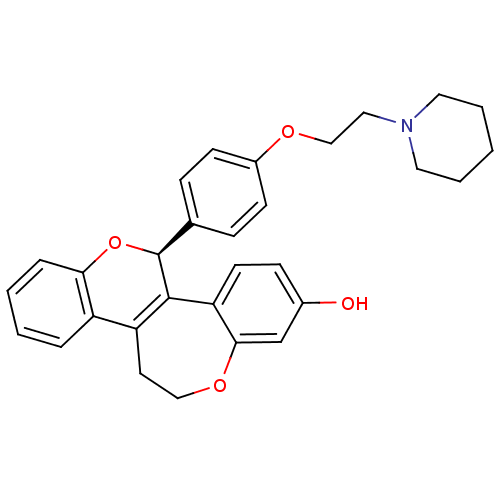
BDBM50310392 5S-(+)-[4-(2-piperidin-1-ylethoxy)phenyl]-11,12-dihydro-5H-6,13-dioxabenzo[3,4]cyclohepta[1,2-a]naphthalen-2-ol::CHEMBL1087426
SMILES Oc1ccc2C3=C(CCOc2c1)c1ccccc1O[C@H]3c1ccc(OCCN2CCCCC2)cc1
InChI Key InChIKey=UHHJAEWQYDXZDY-PMERELPUSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50310392
Found 4 hits for monomerid = 50310392
TargetEstrogen receptor beta(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 24nMAssay Description:Inhibition of fluormone ES2 binding to estrogen receptor beta after 1 hr by fluorescence polarization assayMore data for this Ligand-Target Pair
TargetEstrogen receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 1.24E+3nMAssay Description:Antagonist activity at estrogen receptor in human MCF7 cells assessed as 17beta-estradiol-induced cell proliferation after 24 hrs by [14C]thymidine i...More data for this Ligand-Target Pair
TargetEstrogen receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 150nMAssay Description:Antagonist activity at estrogen receptor in human Ishikawa cells assessed as 17beta-estradiol-induced alkaline phosphatase activity after 3 days by c...More data for this Ligand-Target Pair
TargetEstrogen receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 4.20nMAssay Description:Displacement of radiolabeled estrogen from estrogen receptor alpha by scintillation countingMore data for this Ligand-Target Pair