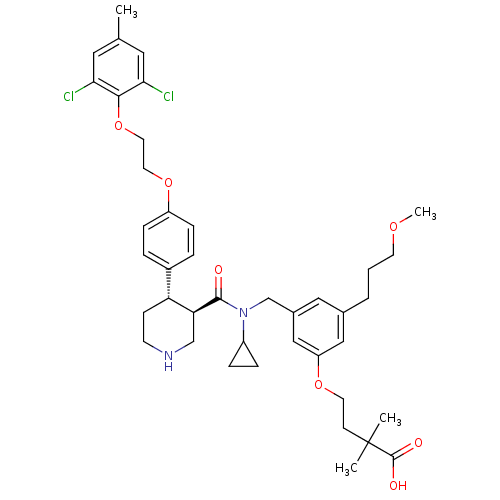
BDBM50340416 4-(3-(((3R,4S)-N-cyclopropyl-4-(4-(2-(2,6-dichloro-4-methylphenoxy)ethoxy)phenyl)piperidine-3-carboxamido)methyl)-5-(3-methoxypropyl)phenoxy)-2,2-dimethylbutanoic acid::CHEMBL1761532
SMILES COCCCc1cc(CN(C2CC2)C(=O)[C@H]2CNCC[C@@H]2c2ccc(OCCOc3c(Cl)cc(C)cc3Cl)cc2)cc(OCCC(C)(C)C(O)=O)c1
InChI Key InChIKey=MVXIVGJQLGJBTD-GPOMZPHUSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50340416
Found 5 hits for monomerid = 50340416
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Merck Frosst Centre For Therapeutic Research
Curated by ChEMBL
Merck Frosst Centre For Therapeutic Research
Curated by ChEMBL
Affinity DataKi: 400nMAssay Description:Binding affinity to human ERGMore data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Inhibition of human recombinant renin in human citrated-plasmaMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Merck Frosst Centre For Therapeutic Research
Curated by ChEMBL
Merck Frosst Centre For Therapeutic Research
Curated by ChEMBL
Affinity DataIC50: >5.00E+4nMAssay Description:Inhibition of human CYP3A4 in human liver microsomes assessed as formation of 6beta-hydroxy-testosterone using testosterone as substrate by high thro...More data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Merck Frosst Centre For Therapeutic Research
Curated by ChEMBL
Merck Frosst Centre For Therapeutic Research
Curated by ChEMBL
Affinity DataIC50: >5.00E+4nMAssay Description:Inhibition of human CYP3A4 in human liver microsomes assessed as formation of 6beta-hydroxy-testosterone using testosterone as substrate by high thro...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0200nMpH: 7.4Assay Description:Inhibition of human recombinant renin in PBS buffer using tetradecapeptide at pH 7.4More data for this Ligand-Target Pair