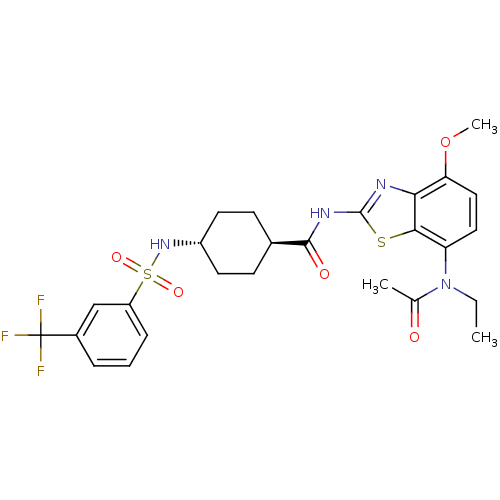
BDBM50341189 CHEMBL1760896::trans-N-(7-(N-ethylacetamido)-4-methoxybenzo[d]thiazol-2-yl)-4-(3-(trifluoromethyl)phenylsulfonamido)cyclohexanecarboxamide
SMILES CCN(C(C)=O)c1ccc(OC)c2nc(NC(=O)[C@H]3CC[C@@H](CC3)NS(=O)(=O)c3cccc(c3)C(F)(F)F)sc12
InChI Key InChIKey=AEALLVSFYNMBHG-SAABIXHNSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50341189
Found 3 hits for monomerid = 50341189
Affinity DataKi: 6nMAssay Description:Displacement of [3H]-DPCPX from human adenosine A1 receptor after 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 200nMAssay Description:Displacement of [3H]ZM241385 from human adenosine A2A receptor after 1 hrMore data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Inhibition of human adenosine A2B receptor expressed in CHO cells assessed as decrease in cellular cAMP level after 20 to 25 minsMore data for this Ligand-Target Pair