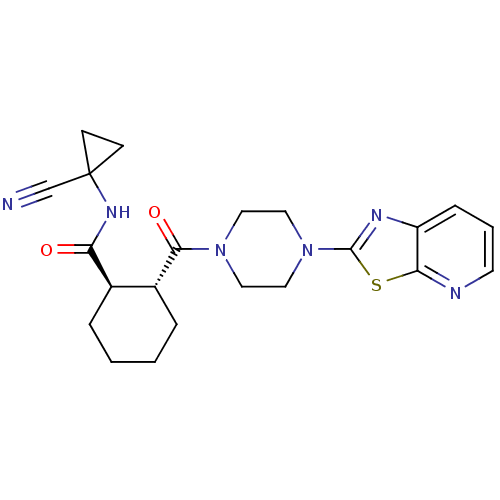
BDBM50390398 CHEMBL2070949
SMILES O=C(NC1(CC1)C#N)[C@@H]1CCCC[C@H]1C(=O)N1CCN(CC1)c1nc2cccnc2s1
InChI Key InChIKey=WIKIUJMLESFHCO-HZPDHXFCSA-N
Data 10 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 10 hits for monomerid = 50390398
Found 10 hits for monomerid = 50390398
Affinity DataIC50: 316nMAssay Description:Inhibition of human recombinant cathepsin K using Z-Phe-Arg-AMC as substrate preincubated for 30 mins measured after 1 hr by quenched fluorescent res...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human recombinant cathepsin L using Z-Phe-Arg-AMC as substrate preincubated for 30 mins measured after 1 hr by quenched fluorescent res...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human recombinant cathepsin S using Z-Val-Val-Arg-AMC as substrate preincubated for 30 mins measured after 1 hr by quenched fluorescent...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human ERGMore data for this Ligand-Target Pair
Affinity DataIC50: 2.51E+4nMAssay Description:Inhibition of human recombinant cathepsin B using Z-Arg-Arg-AMC as substrate preincubated for 30 mins measured after 1 hr by quenched fluorescent res...More data for this Ligand-Target Pair
Affinity DataIC50: >3.16E+4nMAssay Description:Inhibition of CYP2C9More data for this Ligand-Target Pair
Affinity DataIC50: >3.16E+4nMAssay Description:Inhibition of CYP2C19More data for this Ligand-Target Pair
Affinity DataIC50: >3.16E+4nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair
Affinity DataIC50: >3.16E+4nMAssay Description:Inhibition of CYP1A2More data for this Ligand-Target Pair
Affinity DataIC50: >3.16E+4nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair