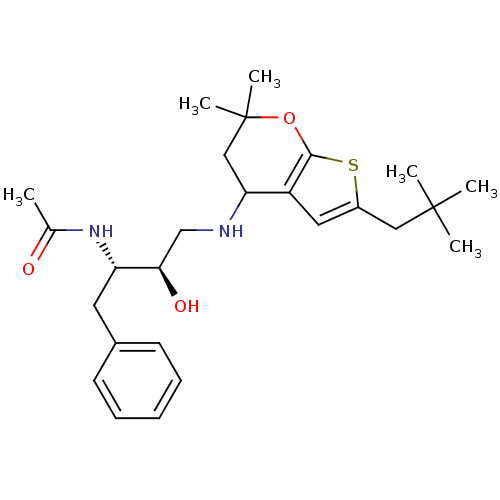
BDBM50393892 CHEMBL2158253
SMILES CC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CNC1CC(C)(C)Oc2sc(CC(C)(C)C)cc12
InChI Key InChIKey=YLIBYDKWVPKMKX-OCESARCHSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50393892
Found 4 hits for monomerid = 50393892
Affinity DataKi: 9.90nMAssay Description:Inhibition of human cathepsin D using DAB-CYL-Glu-ArG-Nle-Phe-Leu-Ser-Phe-Pro-EDANS incubated for 20 mins prior to substrate addition measured after ...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Inhibition of BACE1 in human HEK cells expressing APP Swedish mutant assessed as reduction in amyloid beta40 level by ELISAMore data for this Ligand-Target Pair
Affinity DataIC50: 8.5nMAssay Description:Inhibition of human BACE1 expressed in Escherichia coli BL21(DE3) using Eu-EVNLDAEFK as substrate incubated for 30 mins prior to substrate addition m...More data for this Ligand-Target Pair