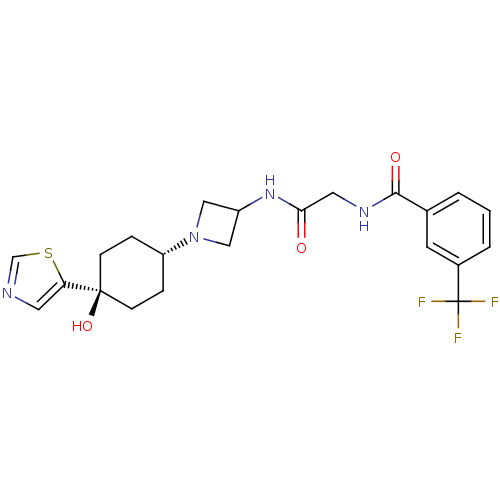
BDBM50400695 CHEMBL2204263
SMILES O[C@]1(CC[C@@H](CC1)N1CC(C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cncs1
InChI Key InChIKey=CFKBNYUHQSQBSX-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50400695
Found 4 hits for monomerid = 50400695
Affinity DataIC50: 30nMAssay Description:Antagonist activity against CCR2 in human THP-1 cells assessed as inhibition of MCP-1 induced chemotaxis after 3 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 37nMAssay Description:Displacement of [125I]-MCP-1 from CCR2 in human THP-1 cells after 2 hrs by microplate scintillation and luminescence counter analysisMore data for this Ligand-Target Pair
Affinity DataKi: 9.60E+3nMAssay Description:Binding affinity to mouse CCR2More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Displacement of [3H]-astemizole from human ERG expressed in HEK293 cells after 1 hr by scintillation counting analysisMore data for this Ligand-Target Pair