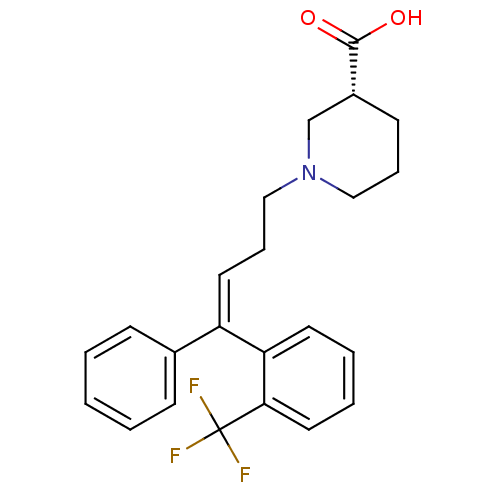
BDBM50417660 CHEMBL1642377
SMILES OC(=O)[C@@H]1CCCN(CC\C=C(\c2ccccc2)c2ccccc2C(F)(F)F)C1
InChI Key InChIKey=YBRSOQNKLWRTMO-GEBKJHSDSA-N
Data 1 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 1 hit for monomerid = 50417660
Found 1 hit for monomerid = 50417660
TargetSodium- and chloride-dependent GABA transporter 1(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataKi: 93.3nMAssay Description:Displacement of [3H]Tiagabine from human recombinant GAT1 expressed in HEK293 cells by equilibrium binding assayMore data for this Ligand-Target Pair