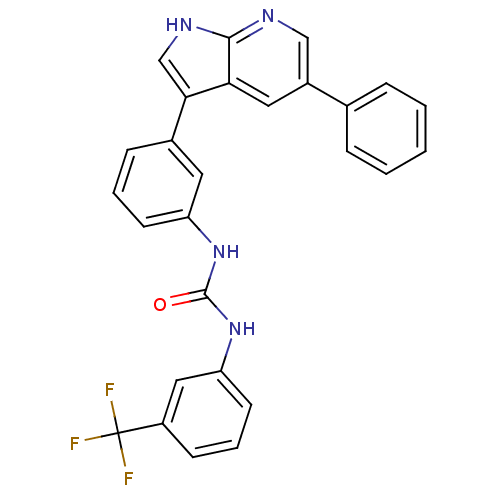
BDBM50446275 CHEMBL3109204
SMILES FC(F)(F)c1cccc(NC(=O)Nc2cccc(c2)-c2c[nH]c3ncc(cc23)-c2ccccc2)c1
InChI Key InChIKey=AMXYLFXYHCZHEV-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50446275
Found 3 hits for monomerid = 50446275
TargetReceptor-interacting serine/threonine-protein kinase 1(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:Displacement of (14-(2-{[3-({2-{[4-(cyanomethyl)phenyl]amino}-6-[(5-cyclopropyl-1H-pyrazol-3-yl)amino]-4-pyrimidinyl}amino)propyl]amino}-2-oxoethyl)-...More data for this Ligand-Target Pair
TargetReceptor-interacting serine/threonine-protein kinase 1(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:Inhibition of human N-terminal His-GST-TEV-fused RIP1 kinase domain (1 to 375) autophosphorylation expressed in baculovirus infected insect Sf9 cells...More data for this Ligand-Target Pair
TargetReceptor-interacting serine/threonine-protein kinase 1(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 79nMAssay Description:Inhibition of RIP1 in human U937 cells assessed as prevention of TNFalpha/zVAD.fmk-induced necrotic cell death preincubated for 30 to 60 mins followe...More data for this Ligand-Target Pair