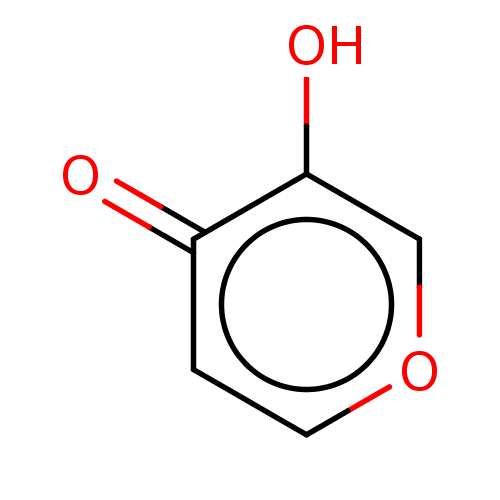
BDBM50536112 CHEMBL79857
SMILES Oc1coccc1=O
InChI Key InChIKey=VEYIMQVTPXPUHA-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50536112
Found 3 hits for monomerid = 50536112
TargetPolymerase acidic protein(Influenza A virus (strain A/Puerto Rico/8/1934 H1N...)
University of California San Diego
Curated by ChEMBL
University of California San Diego
Curated by ChEMBL
Affinity DataIC50: 2.25E+4nMAssay Description:Inhibition of 8-histidine tagged influenza virus H1N1 N-terminal PA endonuclease expressed in Escherichia coli BL21 cells using single-stranded 5'-FA...More data for this Ligand-Target Pair
TargetPolymerase acidic protein(Influenza A virus (strain A/Puerto Rico/8/1934 H1N...)
University of California San Diego
Curated by ChEMBL
University of California San Diego
Curated by ChEMBL
Affinity DataIC50: 2.25E+4nMAssay Description:Inhibition of 8-histidine tagged influenza virus H1N1 N-terminal PA endonuclease expressed in Escherichia coli BL21 cells using single-stranded 5'-FA...More data for this Ligand-Target Pair
Affinity DataIC50: 2.32E+5nMAssay Description:Inhibition of recombinant human QC using H-Gln-AMC hydrobromide as fluorogenic substrate incubated for 6 hrs by fluorometric microplate reader analys...More data for this Ligand-Target Pair