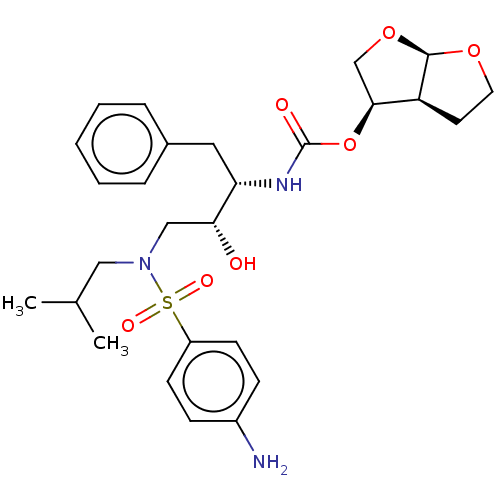
BDBM8125 (3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl N-[(2S,3R)-4-[(4-aminobenzene)(2-methylpropyl)sulfonamido]-3-hydroxy-1-phenylbutan-2-yl]carbamate::CHEMBL1323::Darunavir::Darunavir (DRV)::TMC-114::UIC-94017::US10806794, Compound Darunavir
SMILES CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)O[C@H]1CO[C@H]2OCC[C@@H]12)S(=O)(=O)c1ccc(N)cc1
InChI Key InChIKey=CJBJHOAVZSMMDJ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 101 hits for monomerid = 8125
Found 101 hits for monomerid = 8125
Affinity DataKi: 0.000147nMAssay Description:Inhibition of HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.000200nMAssay Description:Inhibition of wild type HIV1 protease by FRET assayMore data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: <0.00100nMAssay Description:Inhibition of HIV1 recombinant protease L10F/L19I/K20R/L33F/E35D/M36I/R41K/F53L/I54V/L63P/H69K/A71V/T74P/I84V/L89M/L90M/I93L mutant expressed in Esch...More data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.00100nMAssay Description:Inhibition of HIV1 protease I50V, A71V mutant by FRET assayMore data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.00400nMAssay Description:Inhibition of HIV1 protease L10I, G48V, I54V, L63P and V82A mutant by FRET assayMore data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.00430nMAssay Description:Inhibition of HIV1 recombinant protease A71V/V82T/I84V mutant expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
University of Massachusetts Medical School
Curated by ChEMBL
University of Massachusetts Medical School
Curated by ChEMBL
Affinity DataKi: <0.00500nMAssay Description:Inhibition of HIV1 NL4-3 protease expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing protease cleavage ...More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M](Human immunodeficiency virus)
University of Massachusetts
University of Massachusetts
Affinity DataKi: 0.00500nM ΔG°: -15.4kcal/molepH: 4.7 T: 2°CAssay Description:The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper...More data for this Ligand-Target Pair
Affinity DataKi: 0.00500nMAssay Description:Inhibition of wild type HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.00530nMAssay Description:Inhibition of wild-type HIV1 BH10 protease expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.00800nMAssay Description:Binding affinity to wild type HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.00800nMAssay Description:Inhibition of wild type HIV1 protease by FRETMore data for this Ligand-Target Pair
Affinity DataKi: 0.0100nMAssay Description:Inhibition of HIV1 protease using RE(Edans)SGIFLETSK(Dabcyl)R as substrate by fluorescence methodMore data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.0110nMAssay Description:Inhibition of HIV1 recombinant protease D30N/N88D mutant expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0130nMAssay Description:Inhibition of HIV1 protease expressed in Escherichia coli using Val-Ser-Gln-Asn-(beta-naphtyl)Ala-Pro-Ile-Val as substrate preincubated for 30 mins f...More data for this Ligand-Target Pair
TargetDimer of Gag-Pol polyprotein [501-599,Q508K,L534I,L564I,C568A,C596A](Human immunodeficiency virus type 1)
Purdue University
Purdue University
Affinity DataKi: 0.0140nM ΔG°: -14.8kcal/molepH: 6.4 T: 2°CAssay Description:The Ki values were determined by substrate cleavage assay using fluorogenic substrate, 2-(aminobenzoyl)-Thr-Ile-Nle-Phe(p-NO2)-Gln-Arg-NH2. A standar...More data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV-1 protease by fluorometric assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of wild type HIV-1 NL4-3 protease expressed in Escherichia coli Rosetta (DE3)pLysS using Ac-Thr-Ile-Nle-Nle-Gln-Arg-NH2 as substrate by fl...More data for this Ligand-Target Pair
Affinity DataKd: 0.0160nMAssay Description:Binding affinity to HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV1 protease dimerization in MT2 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV1 wild type NL4-3 protease expressed in Escherichia coli Rosetta (DE3) pLysS using Abz-Thr-Ile-Nle-Phe-(pNO2)-Gln-Arg-NH2 as substra...More data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV1 proteaseMore data for this Ligand-Target Pair
TargetDimer of Gag-Pol polyprotein [501-599,Q508K,L534I,L564I,C568A,C596A](Human immunodeficiency virus type 1)
Purdue University
Purdue University
Affinity DataKi: 0.0160nM ΔG°: -14.7kcal/mole EC50: 1.60nMpH: 6.4 T: 2°CAssay Description:The Ki values were determined by substrate cleavage assay using fluorogenic substrate, 2-(aminobenzoyl)-Thr-Ile-Nle-Phe(p-NO2)-Gln-Arg-NH2. A standar...More data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of HIV1 protease using Ac-Thr-Ile-Nle-Nle-Gln-Arg-NH2 substrate by continuous fluorometric assayMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [484-582,Q491K,I497V,V499I,E519D,R541K,D544E,L547P,Q553K,A555V,G557S,I568V,L573M,L574M](Human immunodeficiency virus)
University of Massachusetts
University of Massachusetts
Affinity DataKi: 0.0240nM ΔG°: -14.5kcal/molepH: 4.7 T: 2°CAssay Description:The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper...More data for this Ligand-Target Pair
Affinity DataKi: 0.0250nMAssay Description:Inhibition of HIV1 NL4-3 protease I84V mutant expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing natura...More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [484-582,Q491K,I497V,V499I,E519D,G532V,I538V,R541K,D544E,L547P,Q553K,V566A,L573M](Human immunodeficiency virus)
University of Massachusetts
University of Massachusetts
Affinity DataKi: 0.0250nM ΔG°: -14.5kcal/molepH: 4.7 T: 2°CAssay Description:The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper...More data for this Ligand-Target Pair
Affinity DataKi: 0.0250nMAssay Description:Inhibition of HIV1 NL4-3 protease I84V mutant expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing protea...More data for this Ligand-Target Pair
Affinity DataKi: 0.0260nMAssay Description:Binding affinity to drug-resistant HIV-1 protease I84V mutant expressed in Escherichia coli TAP-106 cells assessed as inhibition constant using a EDA...More data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.0270nMAssay Description:Inhibition of HIV1 protease L10I, L63P, A71V, G73S, I84V, L90M mutant by FRET assayMore data for this Ligand-Target Pair
TargetDimer of Gag-Pol polyprotein [501-599,Q508K,L534I,L564I,C568A,C596A,L591M](Human immunodeficiency virus type 1)
Georgia State University
Georgia State University
Affinity DataKi: 0.0300nMAssay Description:The inhibition assays were performed in microtiter plate wells by mixing enzyme and fluorescent peptide substrate in the presence of inhibitor compou...More data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.0320nMAssay Description:Inhibition of HIV1 recombinant protease M46I/A71V/V82T/I84V mutant expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0600nMAssay Description:Inhibition of HIV1 protease expressed in Escherichia coli using Arg-Glu (EDANS)-Ser-GlnAsn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincuba...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0610nMT: 2°CAssay Description:Inhibition of HIV1 protease expressed in Escherichia coli incubated for 20 to 30 mins at room temperature using (Arg-Glu(EDANS)-Ser-Gln-Asn-Tyr-Pro-I...More data for this Ligand-Target Pair
Affinity DataKi: 0.0750nMAssay Description:Inhibition of HIV1 NL4-3 protease I50V/A71V mutant expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing p...More data for this Ligand-Target Pair
Affinity DataKi: 0.0750nMAssay Description:Binding affinity to drug-resistant HIV-1 protease I50V/A71V mutant expressed in Escherichia coli TAP-106 cells assessed as inhibition constant using ...More data for this Ligand-Target Pair
Affinity DataKi: 0.0750nMAssay Description:Inhibition of HIV1 NL4-3 protease I50V/A71V mutant expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing n...More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-892,N526H,R530K,V566I,I582L,V648I,Q690K,I723V,C750S,T788A,Q795E,T874A,V881I,P882Q](Human immunodeficiency virus)
Massachusetts Institute of Technology
Massachusetts Institute of Technology
Affinity DataEC50: 0.150nMAssay Description:Drug susceptibility assays were carried out by Monogram Bioscience against wild-type HIV-1 control and patient-derived strains of wild-type HIV-1 fro...More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
University of Massachusetts Medical School
Curated by ChEMBL
University of Massachusetts Medical School
Curated by ChEMBL
Affinity DataKi: 0.180nMAssay Description:Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi...More data for this Ligand-Target Pair
TargetHIV WT-C pol protein (wild-type clade C)(Human immunodeficiency virus)
Massachusetts Institute of Technology
Massachusetts Institute of Technology
Affinity DataEC50: 0.180nMAssay Description:Drug susceptibility assays were carried out by Monogram Bioscience against wild-type HIV-1 control and patient-derived strains of wild-type HIV-1 fro...More data for this Ligand-Target Pair
TargetDimer of Gag-Pol polyprotein [501-599,Q508K,L534I,L564I,C568A,C596A](Human immunodeficiency virus type 1)
Georgia State University
Georgia State University
Affinity DataKi: 0.220nM ΔG°: -13.7kcal/molepH: 5.6 T: 2°CAssay Description:The inhibition assays were performed in microtiter plate wells by mixing enzyme and fluorescent peptide substrate in the presence of inhibitor compou...More data for this Ligand-Target Pair
Affinity DataEC50: 0.230nMAssay Description:Drug susceptibility assays were carried out by Monogram Bioscience against wild-type HIV-1 control and patient-derived strains of wild-type HIV-1 fro...More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,I534V,R541K,D544E,Q553K,A555V,L573M](Human immunodeficiency virus)
University of Massachusetts
University of Massachusetts
Affinity DataKi: 0.245nM ΔG°: -13.1kcal/molepH: 4.7 T: 2°CAssay Description:The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper...More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-892](Human immunodeficiency virus)
Massachusetts Institute of Technology
Massachusetts Institute of Technology
Affinity DataEC50: 0.25nMAssay Description:Drug susceptibility assays were carried out by Monogram Bioscience against wild-type HIV-1 control and patient-derived strains of wild-type HIV-1 fro...More data for this Ligand-Target Pair
TargetHIV WT-A pol protein (wild-type clade A)(Human immunodeficiency virus type 1)
Massachusetts Institute of Technology
Massachusetts Institute of Technology
Affinity DataEC50: 0.300nMAssay Description:Drug susceptibility assays were carried out by Monogram Bioscience against wild-type HIV-1 control and patient-derived strains of wild-type HIV-1 fro...More data for this Ligand-Target Pair
TargetHIV-1 protease(Human immunodeficiency virus)
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Academy of Sciences of The Czech Republic
Curated by ChEMBL
Affinity DataKi: 0.310nMAssay Description:Inhibition of HIV1 recombinant protease V32I/I47A mutant expressed in Escherichia coli by spectrophotometric assayMore data for this Ligand-Target Pair