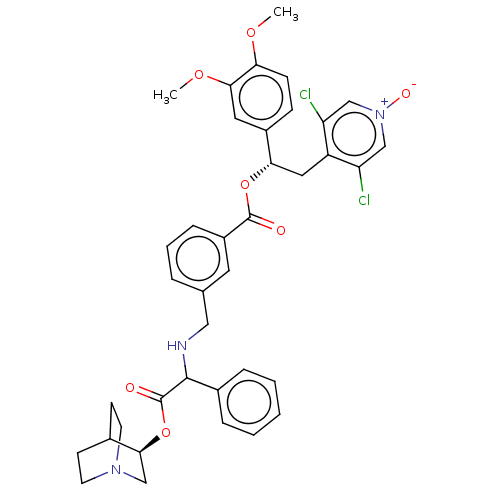
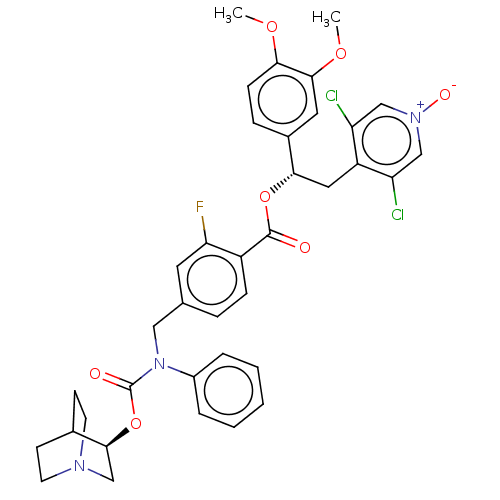
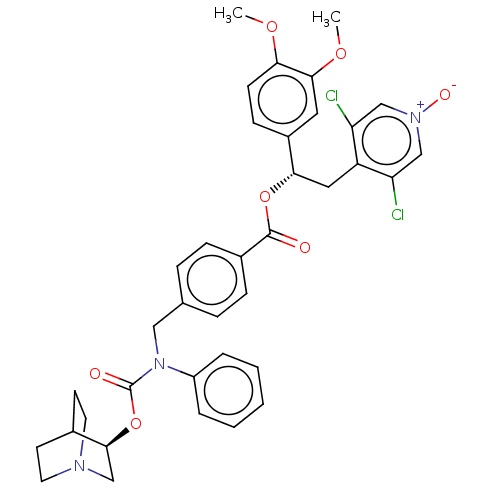
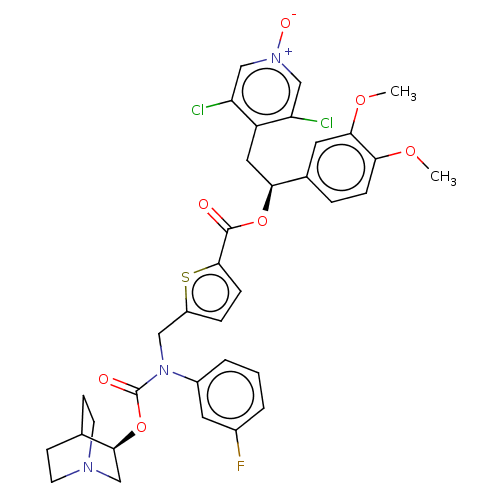
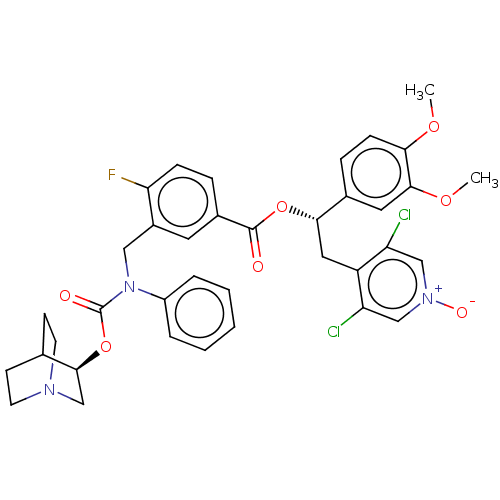
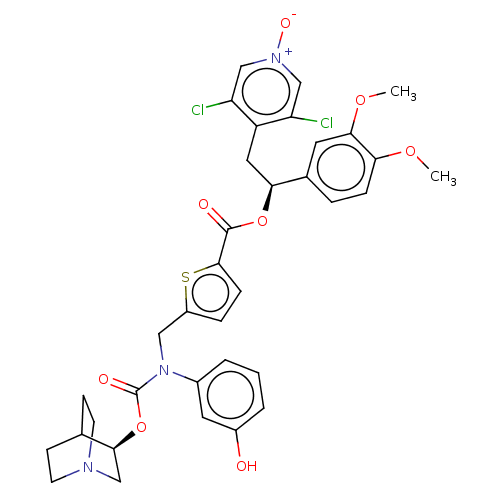
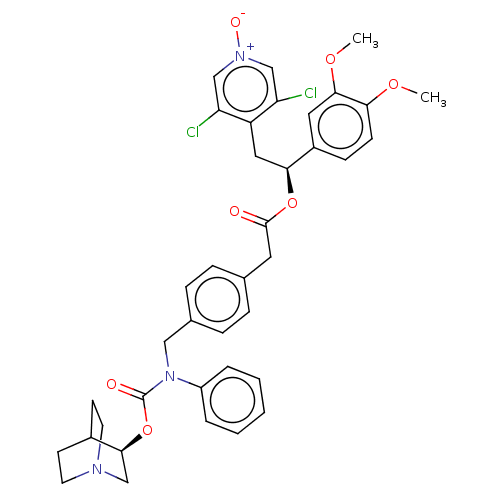
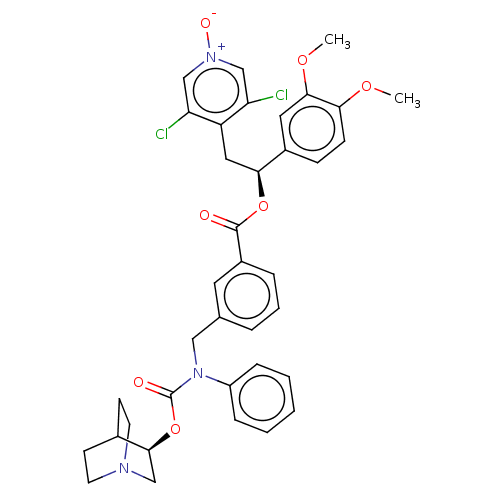
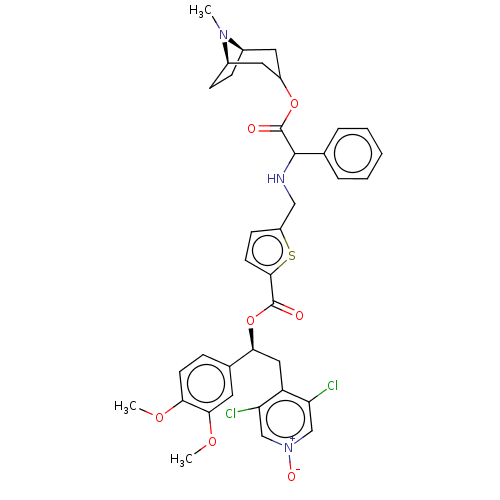
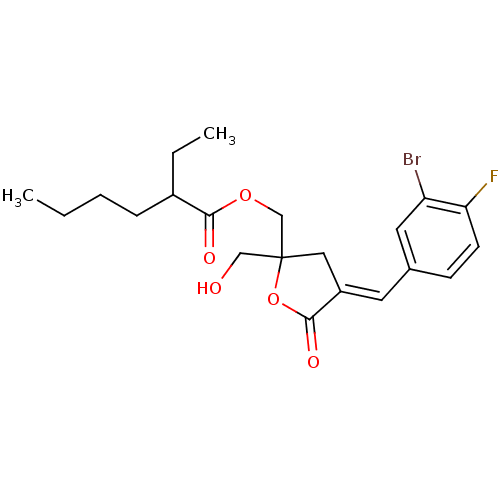
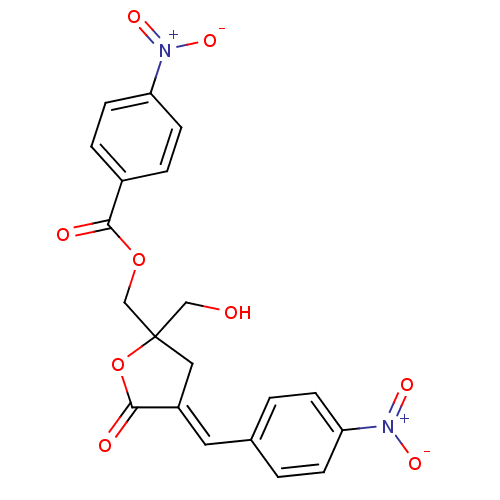
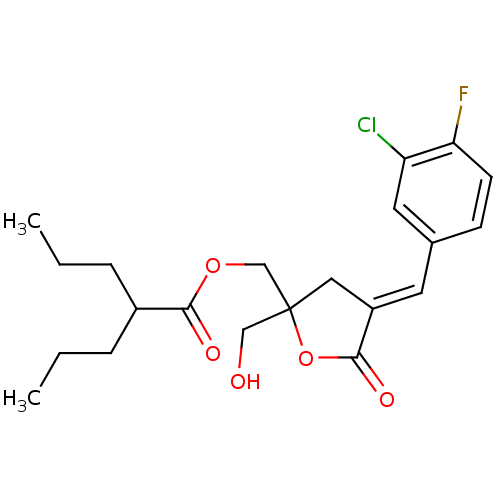
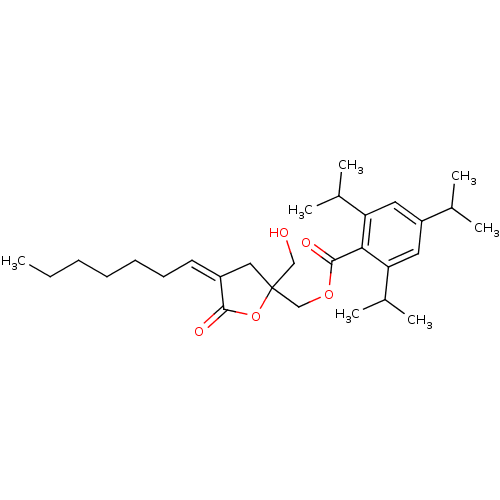
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.100nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
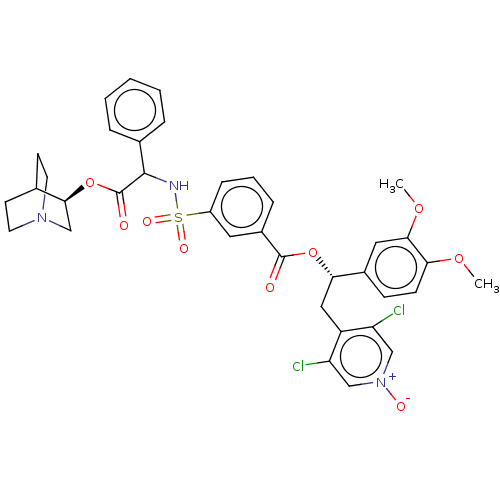
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.100nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
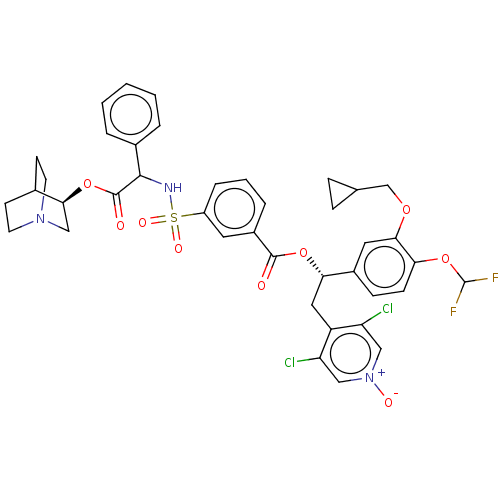
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.100nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
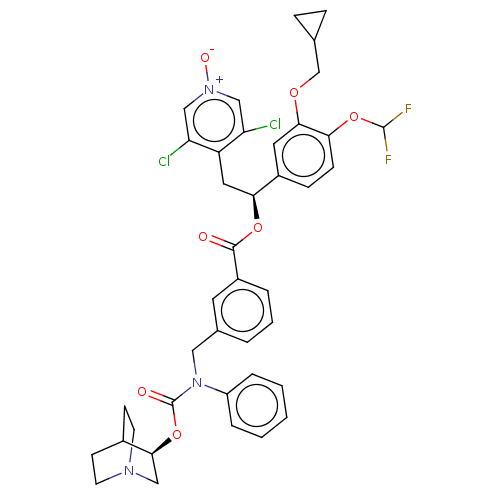
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.126nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.126nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.126nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.126nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.158nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M2(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.158nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M2 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.158nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.158nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M2(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M2 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.251nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.316nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.316nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.316nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.316nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.316nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.398nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.398nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.398nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.501nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.501nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.501nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.631nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.631nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.631nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.631nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.794nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.794nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M2(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.794nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M2 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 0.794nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetTYR_PHOSPHATASE_2 domain-containing protein(Mycobacterium tuberculosis)
Purdue University
Curated by ChEMBL
Purdue University
Curated by ChEMBL
Affinity DataKi: 1.20nMAssay Description:Competitive inhibition of Mycobacterium tuberculosis PTPB expressed in Escherichia coli BL21 (DE3) using varying levels of p-nitrophenyl phosphate as...More data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
TargetRas guanyl-releasing protein 3(Homo sapiens (Human))
National Cancer Institute-Frederick
Curated by ChEMBL
National Cancer Institute-Frederick
Curated by ChEMBL
Affinity DataKi: 1.80nMAssay Description:Binding affinity to RasGRP3 (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.07nMAssay Description:Displacement of [3H]PDBu form mouse PKCalpha by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 2.07nMAssay Description:Displacement of [3H]PDBu form mouse PKCalpha by scintillation countingMore data for this Ligand-Target Pair
TargetRas guanyl-releasing protein 3(Homo sapiens (Human))
National Cancer Institute-Frederick
Curated by ChEMBL
National Cancer Institute-Frederick
Curated by ChEMBL
Affinity DataKi: 2.22nMAssay Description:Binding affinity to RasGRP3 (unknown origin)More data for this Ligand-Target Pair
TargetRas guanyl-releasing protein 3(Homo sapiens (Human))
National Cancer Institute-Frederick
Curated by ChEMBL
National Cancer Institute-Frederick
Curated by ChEMBL
Affinity DataKi: 2.30nMAssay Description:Binding affinity to RasGRP3 (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.45nMAssay Description:Displacement of [3H]PDBu form mouse PKCalpha by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 2.45nMAssay Description:Displacement of [3H]PDBu form mouse PKCalpha by scintillation countingMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 2.5nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 2.5nMAssay Description:Displacement of [3H]PDBu form mouse PKCalpha by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 2.5nMAssay Description:Displacement of [3H]PDBu form mouse PKCalpha by scintillation countingMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(Homo sapiens (Human))
Chiesi Farmaceutici
Curated by ChEMBL
Chiesi Farmaceutici
Curated by ChEMBL
Affinity DataKi: 2.51nMAssay Description:Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 2.66nMAssay Description:Displacement of [3H]PDBu form mouse PKCalpha by scintillation countingMore data for this Ligand-Target Pair