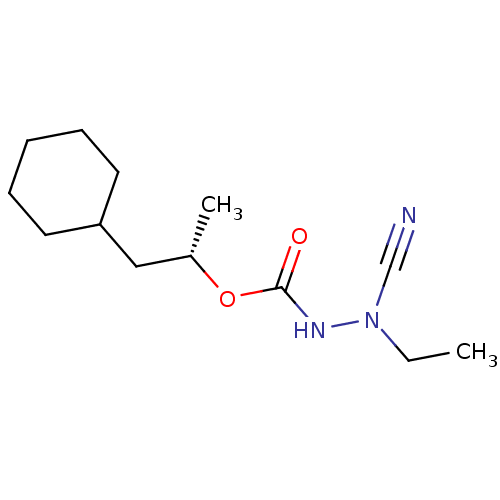
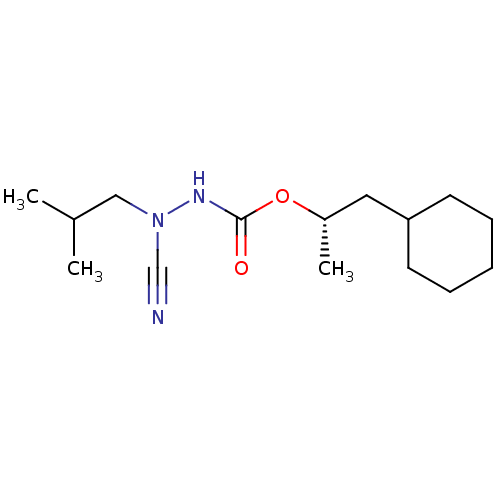
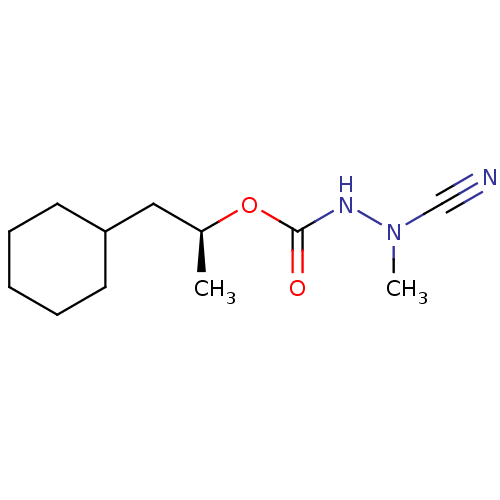
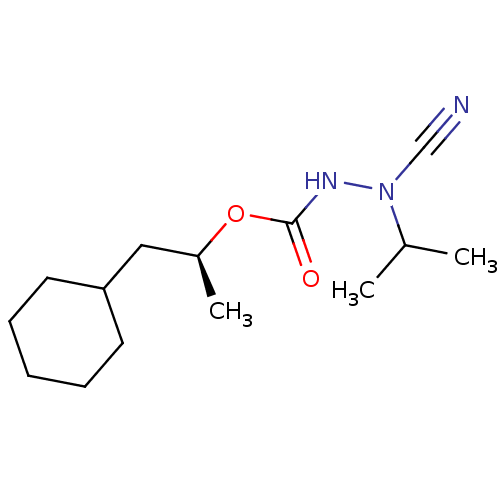
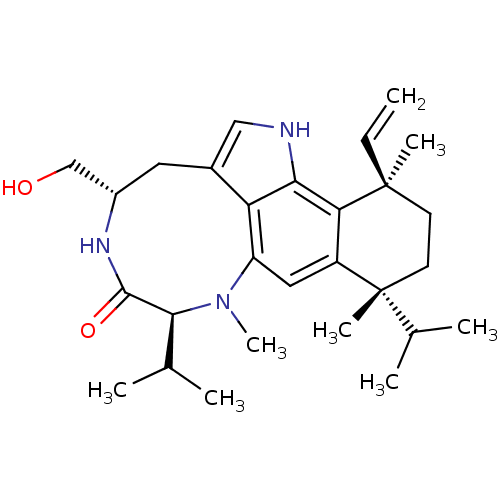
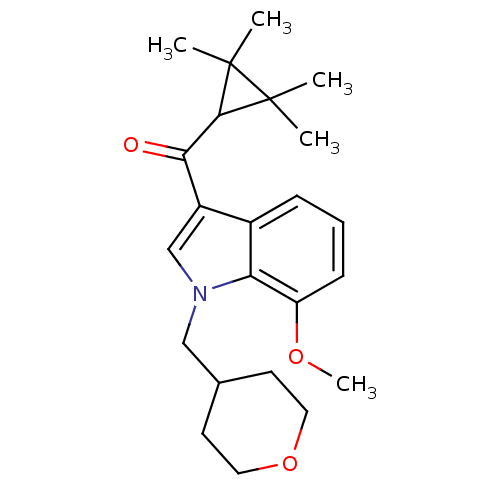
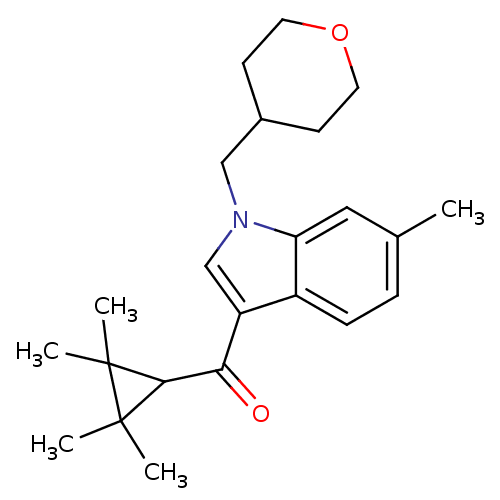
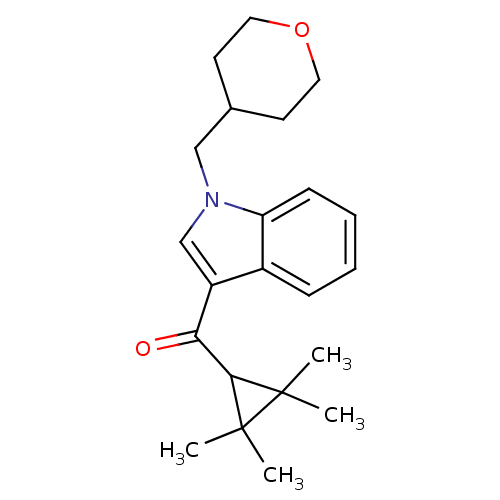
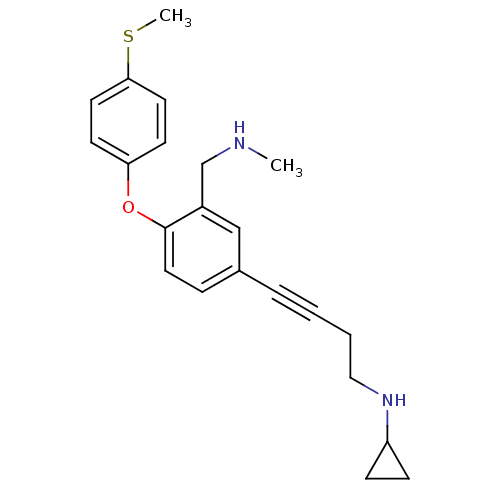
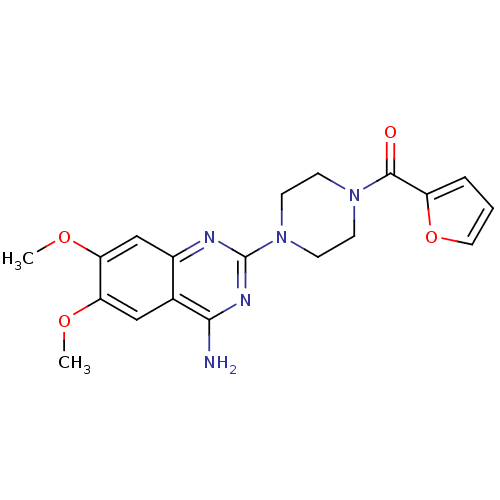
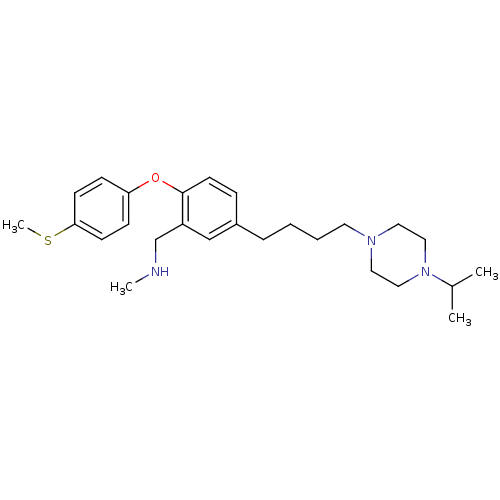
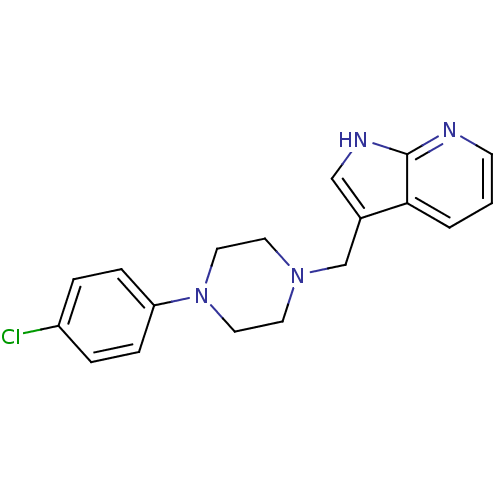
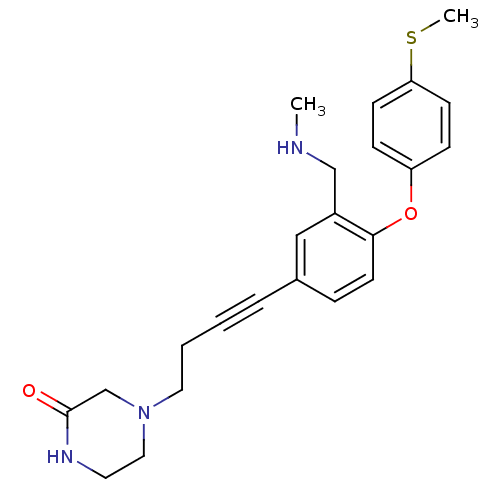
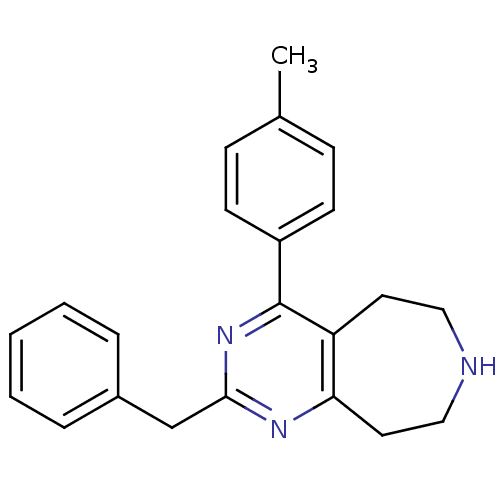
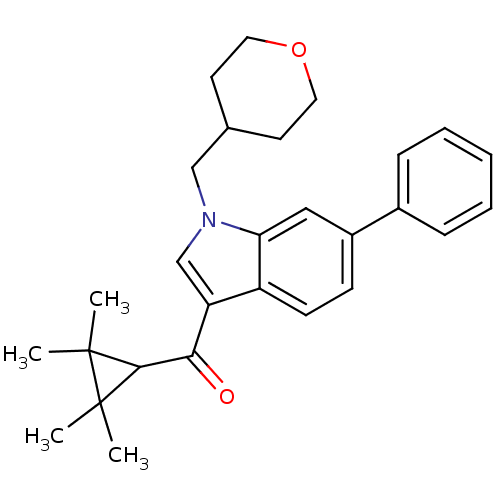
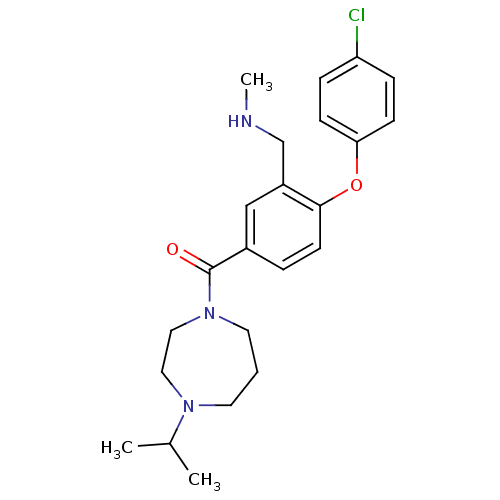
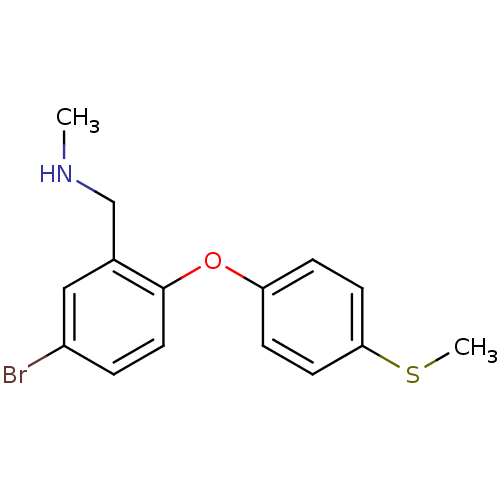
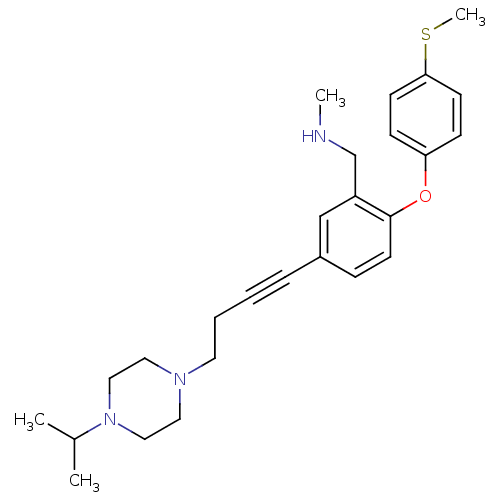
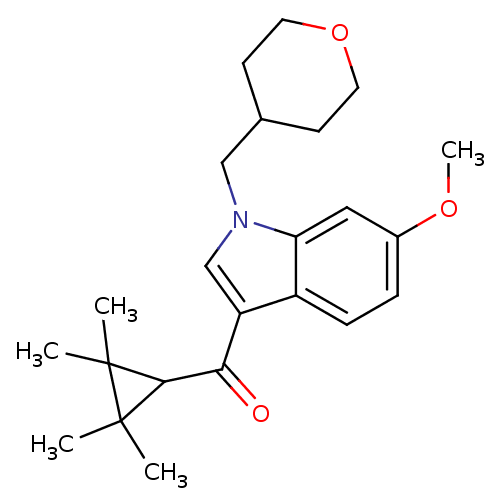
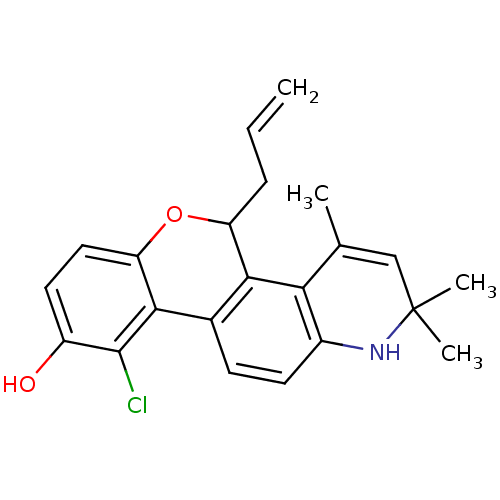
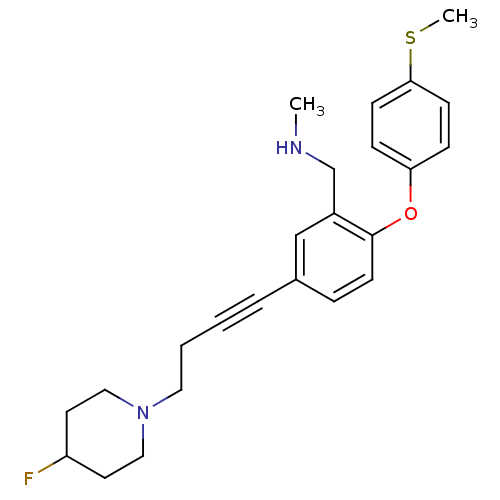
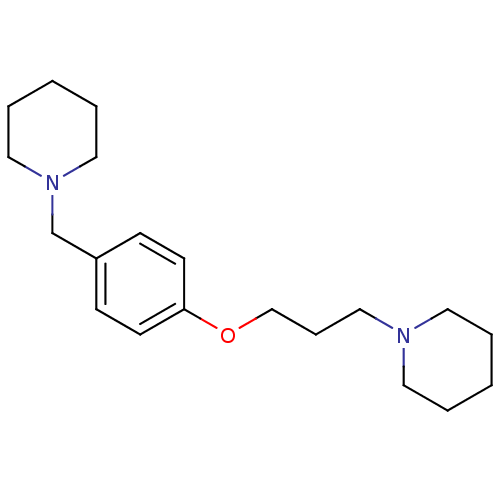
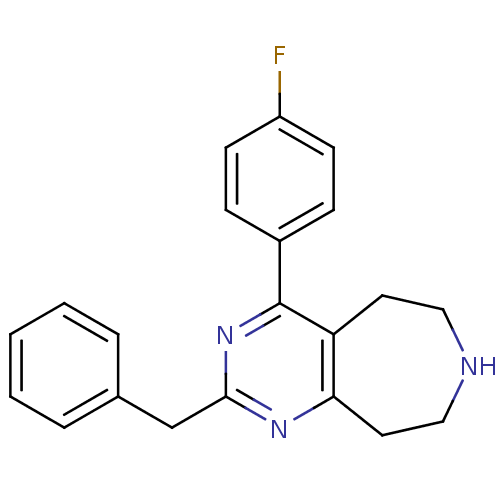
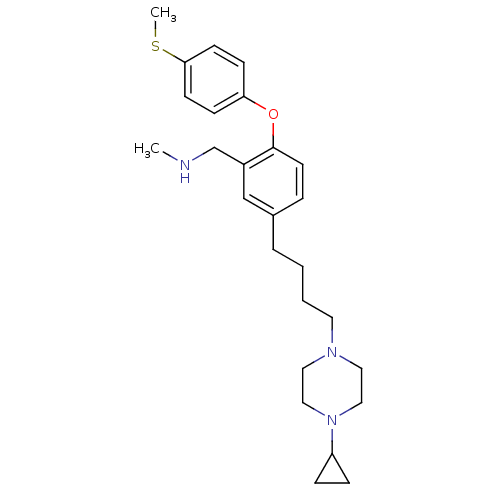
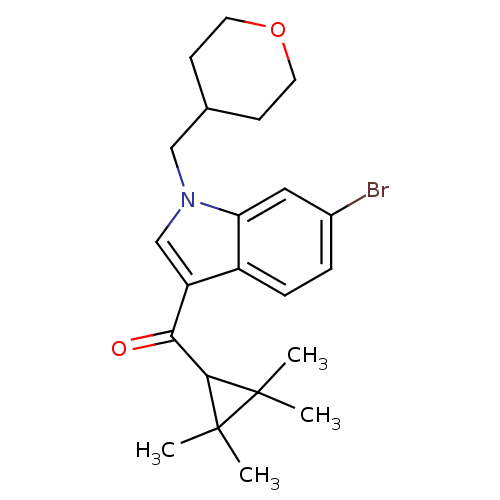
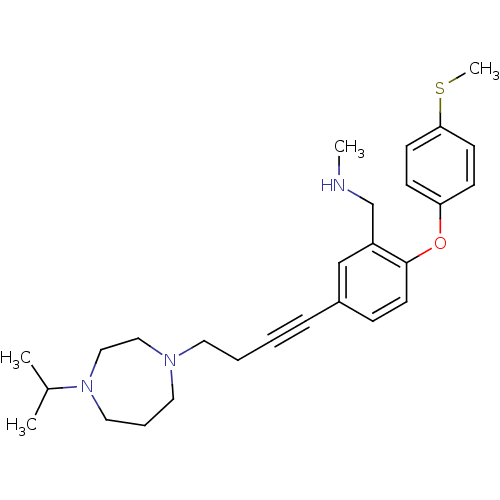
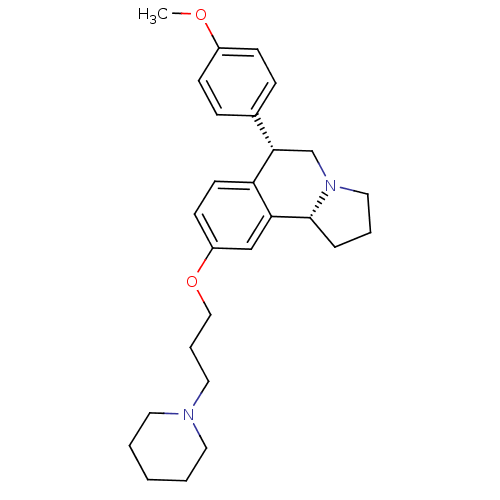
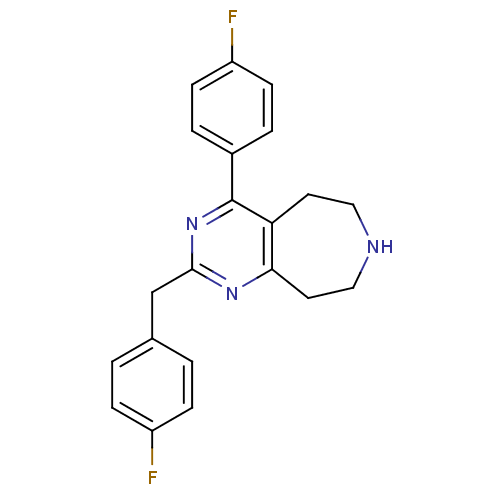
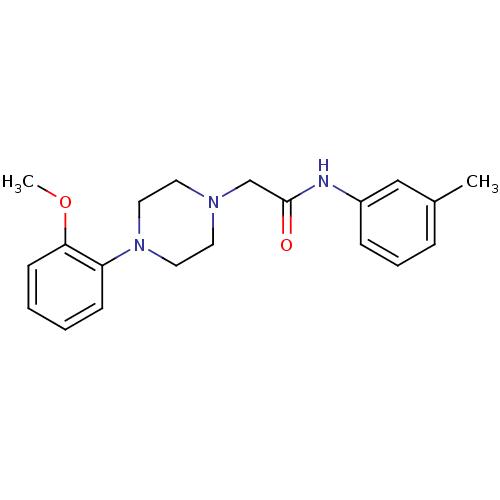
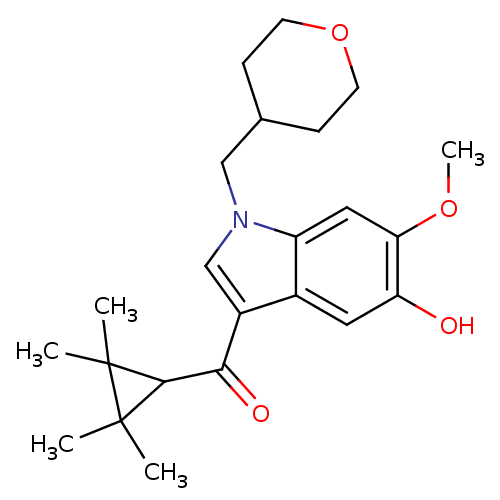
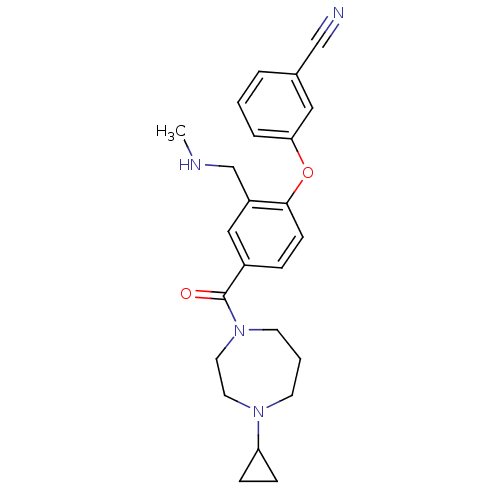
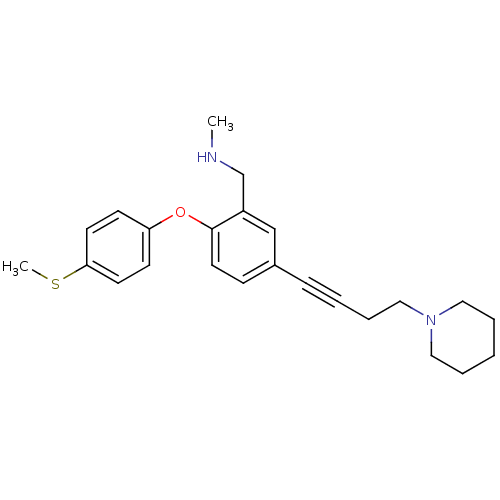
Affinity DataKi: 0.00300nMAssay Description:Inhibition constant against human cathepsin K in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.00400nMAssay Description:Inhibition constant against human cathepsin K in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.00400nMAssay Description:Inhibition constant against human cathepsin K in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.00600nMAssay Description:Inhibition constant against human cathepsin K in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.00700nMAssay Description:Inhibition constant against human cathepsin K in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.00900nMAssay Description:Inhibition constant against human cathepsin K in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.0190nMAssay Description:Inhibition constant against human cathepsin K in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.0250nMAssay Description:Inhibition constant against human cathepsin L using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.0320nMAssay Description:Inhibition constant against human cathepsin K in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.0360nMAssay Description:Inhibition constant against human cathepsin L using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.0720nMAssay Description:Inhibition constant against rat cathepsin KMore data for this Ligand-Target Pair
Affinity DataKi: 0.0770nMAssay Description:Inhibition constant against human cathepsin B in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.100nMAssay Description:Inhibition of [3H]- PDBu binding to peptide D of mouse skin Protein kinase C etaMore data for this Ligand-Target Pair
Affinity DataKi: 0.120nM EC50: >1.00E+4nMAssay Description:IC50 values for test compounds were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibition c...More data for this Ligand-Target Pair
Affinity DataKi: 0.150nM EC50: >1.00E+4nMAssay Description:IC50 values for test compounds were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibition c...More data for this Ligand-Target Pair
Affinity DataKi: 0.210nM EC50: 9.5nMAssay Description:IC50 values for test compounds were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibition c...More data for this Ligand-Target Pair
Affinity DataKi: 0.25nMAssay Description:Inhibition constant against human cathepsin B in fluorescence assay using Cbz-Phe-Arg-AMCMore data for this Ligand-Target Pair
Affinity DataKi: 0.280nMAssay Description:The purpose of CDK2/Cyclin E1 assay is to evaluate the inhibition (% inhibition, Kiapp and Ki values) of small molecule inhibitors by using a fluores...More data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.300nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetAlpha-1A/Alpha-1B/Alpha-1D adrenergic receptor(Rattus norvegicus (rat))
Combichem
Curated by ChEMBL
Combichem
Curated by ChEMBL
Affinity DataKi: 0.320nMAssay Description:Evaluated for binding affinity against alpha-1 adrenergic receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.400nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:Binding affinity against human Dopamine receptor D4 by [3H]-spiperone displacement.More data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.400nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 0.400nMAssay Description:Displacement of [3H]ketanserin from human recombinant 5HT2A receptor expressed in mouse NIH3T3 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.410nM EC50: >1.00E+4nMAssay Description:IC50 values for test compounds were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibition c...More data for this Ligand-Target Pair
Affinity DataKi: 0.460nMAssay Description:Inhibition constant against human cathepsin H using L-Arg-b-naphthalamideMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:Inhibition of human histamine H3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.510nM EC50: >1.00E+4nMAssay Description:IC50 values for test compounds were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibition c...More data for this Ligand-Target Pair
TargetAlpha-1A/Alpha-1B/Alpha-1D adrenergic receptor(Rattus norvegicus (rat))
Combichem
Curated by ChEMBL
Combichem
Curated by ChEMBL
Affinity DataKi: 0.540nMAssay Description:Evaluated for binding affinity against alpha-1 adrenergic receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.550nMAssay Description:The purpose of CDK2/Cyclin E1 assay is to evaluate the inhibition (% inhibition, Kiapp and Ki values) of small molecule inhibitors by using a fluores...More data for this Ligand-Target Pair
Affinity DataKi: 0.580nMAssay Description:Binding affinity towards human glucocorticoid receptor (GR) was determined using [3H]-Dexamethasone as radioligand in SF-1 cellsMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Binding affinity to human histamine H3 receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Displacement of [3H]ketanserin from human recombinant 5HT2A receptor expressed in mouse NIH3T3 cellsMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.600nMAssay Description:Inhibition of [3H]- PDBu binding to peptide D of mouse skin Protein kinase C etaMore data for this Ligand-Target Pair
Affinity DataKi: 0.600nMAssay Description:Affinity for Adenosine A1 receptor determined by [3H]N6-cyclohexyladenosine binding to rat brain membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.650nM EC50: >1.00E+4nMAssay Description:IC50 values for test compounds were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibition c...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Homo sapiens (Human))
Glaxo Wellcome Research And Development
Curated by ChEMBL
Glaxo Wellcome Research And Development
Curated by ChEMBL
Affinity DataKi: 0.692nMAssay Description:Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gammaMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Inhibition of human histamine H3 receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Displacement of [3H]ketanserin from human recombinant 5HT2A receptor expressed in mouse NIH3T3 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:In vitro ability to inhibit [3H]-spiperone binding to human Dopamine receptor D4.4 alleleMore data for this Ligand-Target Pair
Affinity DataKi: 0.700nM EC50: >1.00E+4nMAssay Description:IC50 values for test compounds were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibition c...More data for this Ligand-Target Pair
TargetHistamine H3 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Inhibition of human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity to human histamine H3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity to human SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair