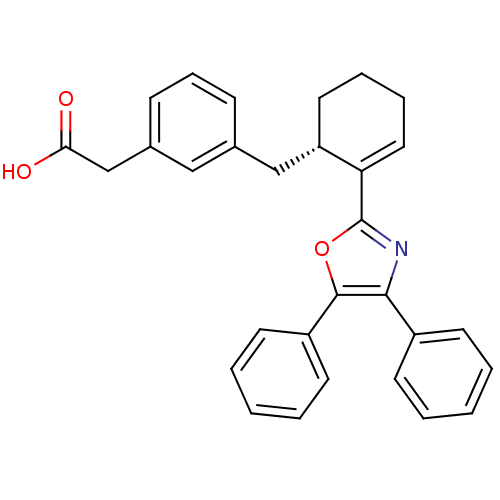
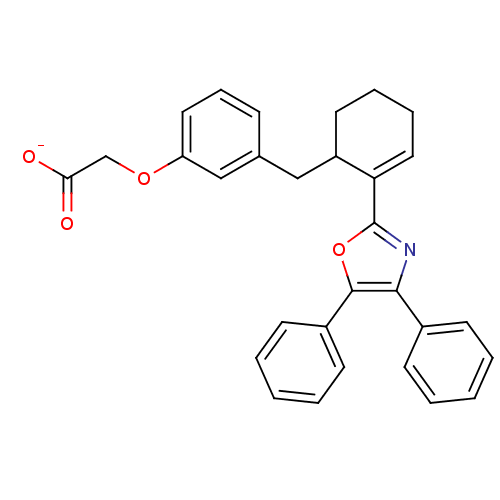
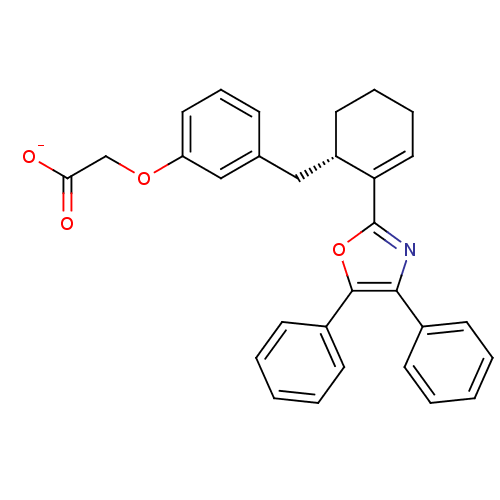
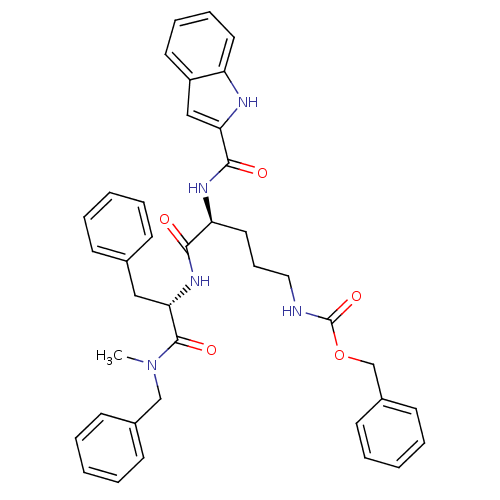
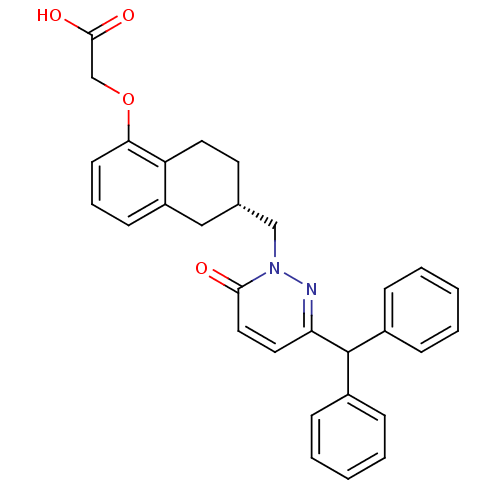
Affinity DataKi: 0.0330nM ΔG°: -59.2kJ/molepH: 7.4 T: 2°CAssay Description:The reaction velocity was measured by change in absorbance at 265nm (A265) resulting from the deamination of adenosine. The reaction was started by a...More data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 0.300nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 0.680nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Rattus norvegicus)
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 0.740nMAssay Description:Inhibition of rat prostanoid EP4 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 0.910nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Rattus norvegicus)
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 5.10nMAssay Description:Inhibition of rat prostanoid EP4 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 5.5nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 6.10nMAssay Description:Inhibition of [3H]-Iloprost binding to human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 6.5nMAssay Description:Ability to inhibit binding of [3H]iloprost to cloned human prostaglandin I2 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 8.40nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 8.90nMAssay Description:Displacement of [3H]iloprost from human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Displacement of [3H]iloprost from human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Ability to inhibit binding of [3H]iloprost to cloned human prostaglandin I2 receptorMore data for this Ligand-Target Pair
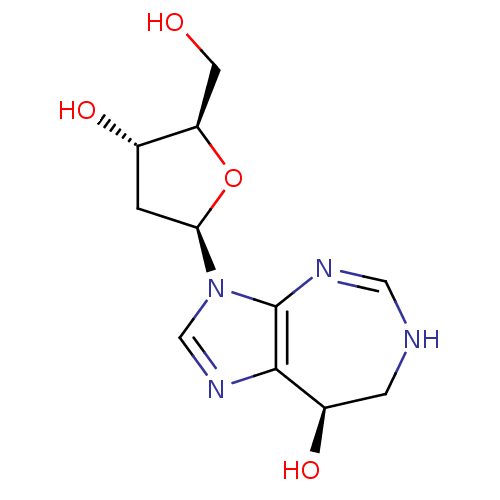
Affinity DataKi: 37nM ΔG°: -42.0kJ/molepH: 7.4 T: 2°CAssay Description:The reaction velocity was measured by change in absorbance at 265nm (A265) resulting from the deamination of adenosine. The reaction was started by a...More data for this Ligand-Target Pair
Affinity DataKi: 41nMAssay Description:Inhibition of [3H]-Iloprost binding to human Prostanoid IP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 43nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 50nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 54nMAssay Description:Inhibition of [3H]-Iloprost binding to human prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 54nMAssay Description:Inhibition of [3H]-Iloprost binding to human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 54nMAssay Description:Displacement of [3H]iloprost from human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 54nMAssay Description:Ability to inhibit binding of [3H]iloprost to cloned human prostaglandin I2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 80nMAssay Description:Tested for inhibition of 3[H]-iloprost binding to human IP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 80nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 94nMAssay Description:Tested for inhibition of 3[H]-iloprost binding to human IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 110nMAssay Description:Displacement of [3H]iloprost from human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:Displacement of [3H]iloprost from human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 170nMAssay Description:Inhibition of [3H]-Iloprost binding to human prostanoid IP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP4 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: 240nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP4 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 610nMAssay Description:Inhibition of [3H]-Iloprost binding to human prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 930nMAssay Description:Inhibition of [3H]-SQ-29,548 binding to human Prostanoid TP receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGF-2 binding to human prostanoid FP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP1 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP1 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP1 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP1 receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP3 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP3 receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-SQ-29,548 binding to human prostanoid TP receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibtion of [3H]-PGD-2 binding to human prostanoid DP receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-Iloprost binding to human prostanoid IP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP2 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGF-2 binding to human prostanoid FP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP2 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-SQ-29,548 binding to human prostanoid TP receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibtion of [3H]-PGD-2 binding to human prostanoid DP receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGF-2 binding to human prostanoid FP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP3 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of [3H]-PGE-2 binding to human prostanoid EP3 receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]PGD-2 from human Prostanoid DP receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]SQ-29,548 from human Prostanoid TP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP1 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]PGE-2 from human Prostanoid EP1 receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]PGD-2 from human Prostanoid DP receptorMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP2 subtype(Homo sapiens (Human))
Fujisawa Pharmaceutical
Curated by ChEMBL
Fujisawa Pharmaceutical
Curated by ChEMBL
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]-PGE-2 from human Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]PGF-2 from human Prostanoid FP receptorMore data for this Ligand-Target Pair






 3D Structure (crystal)
3D Structure (crystal)