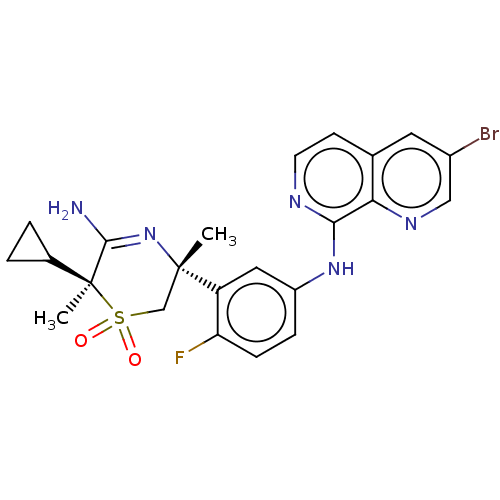
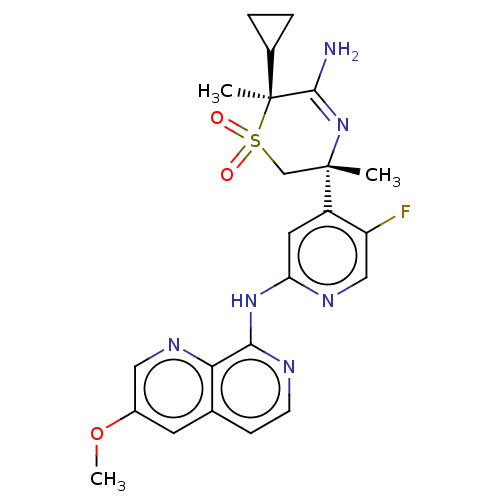
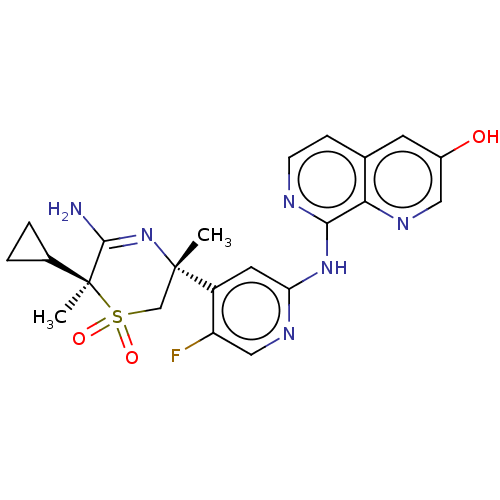
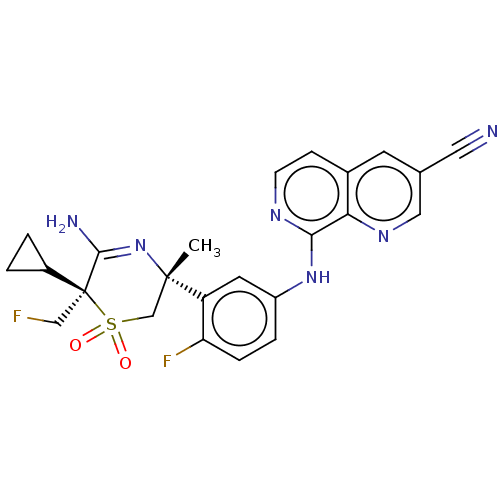
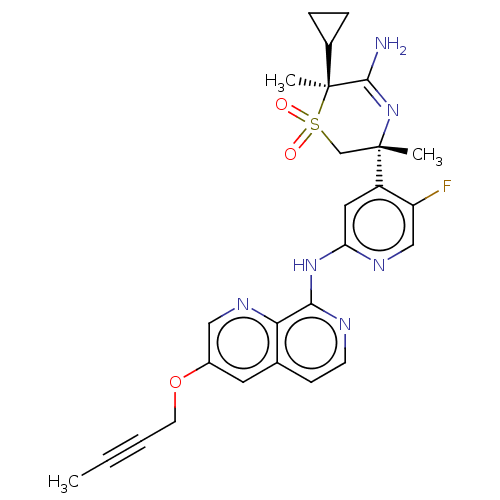
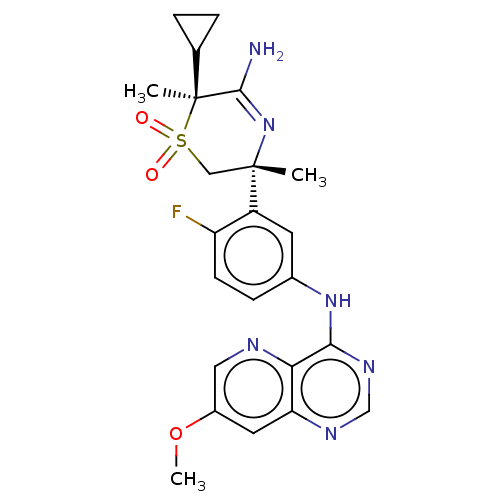
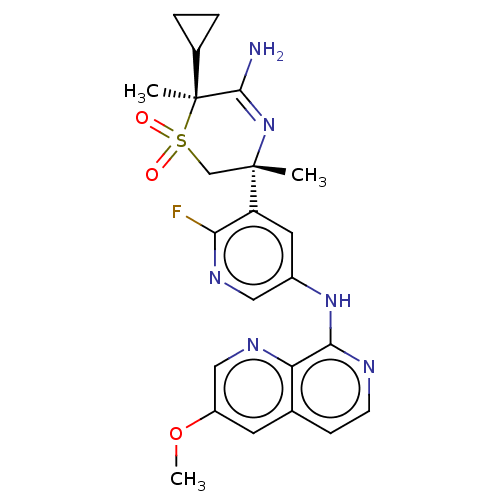
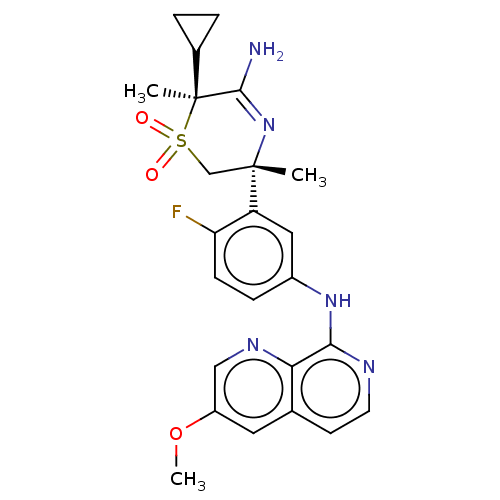
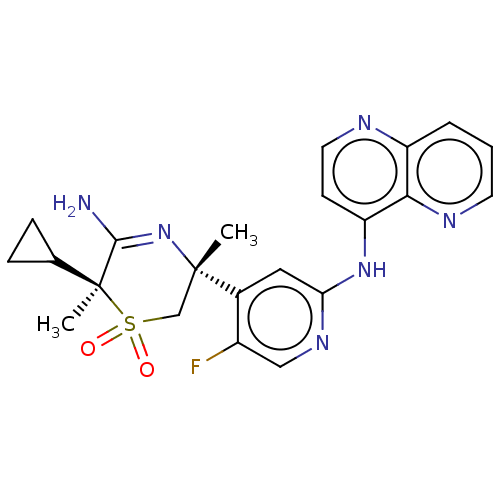
Affinity DataKi: 0.350nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
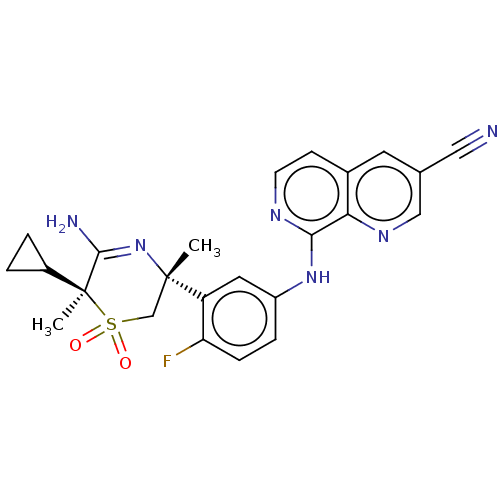
Affinity DataKi: 0.410nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
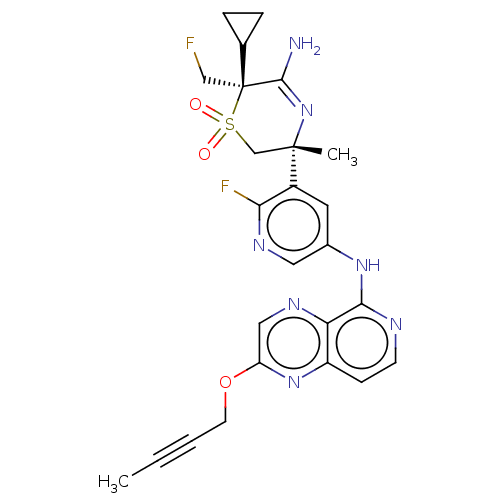
Affinity DataKi: 0.570nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
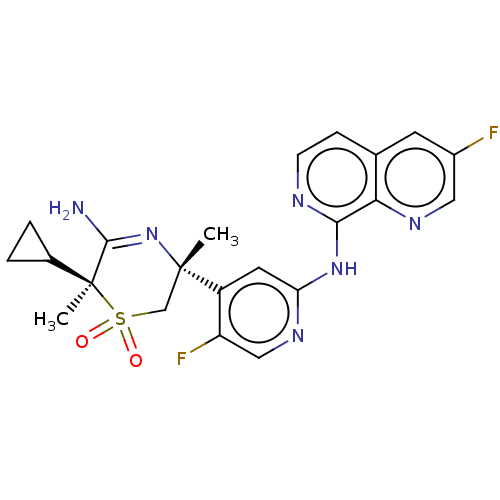
Affinity DataKi: 0.590nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 0.600nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 0.640nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
TargetSomatostatin receptor type 3(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 0.640nMAssay Description:Displacement of [125I]SS14 from human SST3 expressed in CHO membrane after 60 to 90 mins by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 0.640nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 0.650nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 0.730nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 0.790nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
TargetSomatostatin receptor type 3(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 0.815nMAssay Description:Displacement of [125I]SS14 from human SST3 expressed in CHO membrane after 60 to 90 mins by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 0.820nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 0.890nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 0.910nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 1.70nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 2.40nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 2.80nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 2.80nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 2.90nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
TargetSomatostatin receptor type 3(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Displacement of [125I]SS14 from human SST3 expressed in CHO membrane after 60 to 90 mins by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 3.30nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 3.5nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 3.60nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 3.60nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
TargetSomatostatin receptor type 3(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 3.70nMAssay Description:Displacement of [125I]SS14 from human SST3 expressed in CHO membrane after 60 to 90 mins by scintillation countingMore data for this Ligand-Target Pair
TargetSomatostatin receptor type 3(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 3.80nMAssay Description:Displacement of [125I]SS14 from human SST3 expressed in CHO membrane after 60 to 90 mins by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 3.80nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-1 using the following assay.The following reagents were used in this a...More data for this Ligand-Target Pair
Affinity DataKi: 3.80nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair
TargetSomatostatin receptor type 3(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 4.20nMAssay Description:Displacement of [125I]SS14 from human SST3 expressed in CHO membrane after 60 to 90 mins by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 4.20nMAssay Description:The compounds of the invention were determined to be potent inhibitors of BACE-2 using the following assay. Inhibitor IC50s at purified human autoBAC...More data for this Ligand-Target Pair