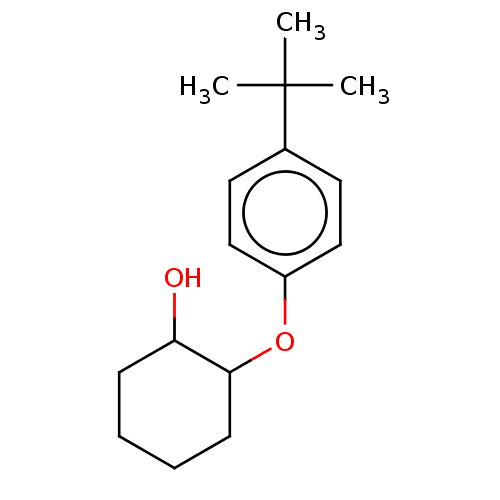
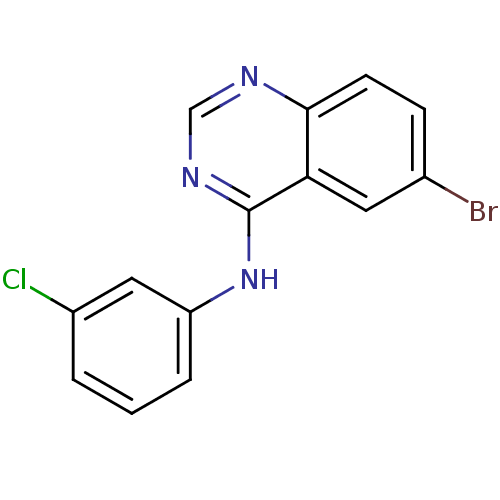
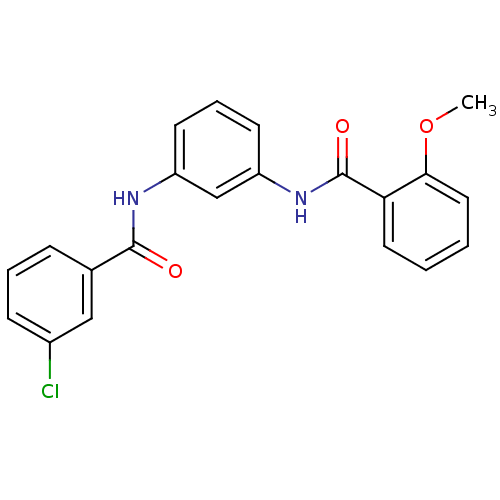
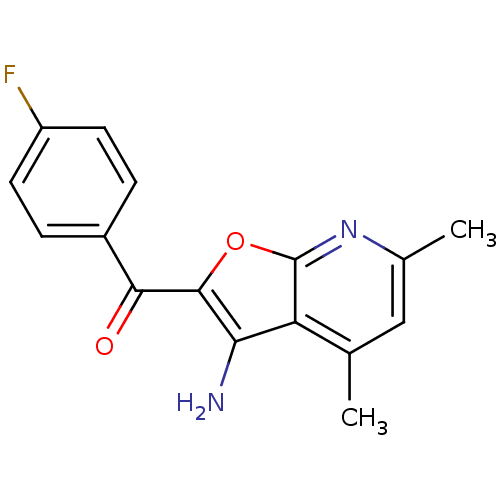
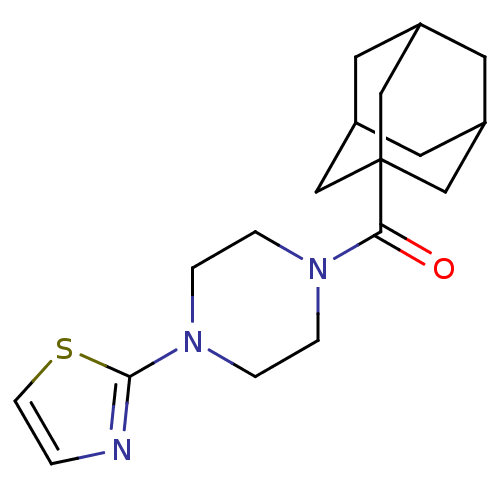
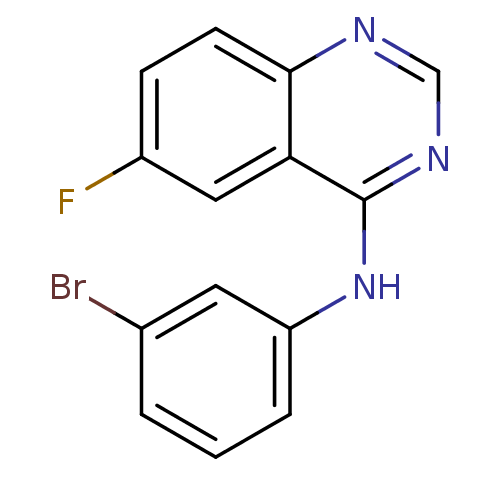
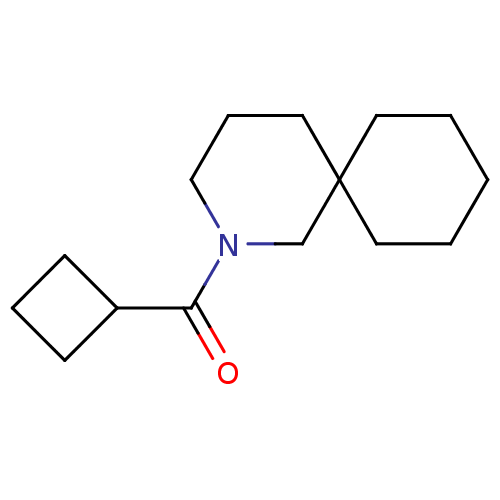
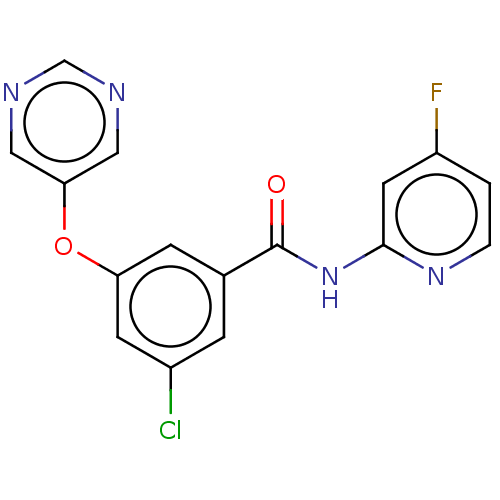
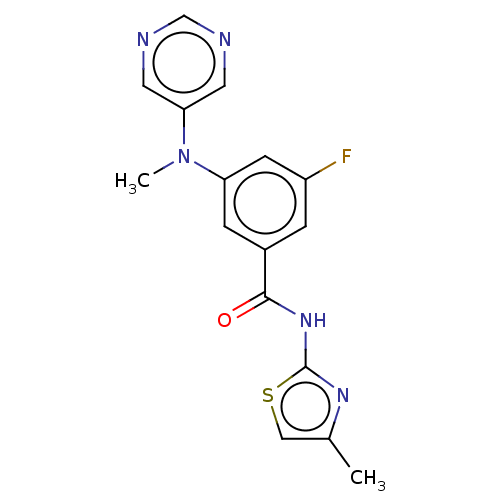
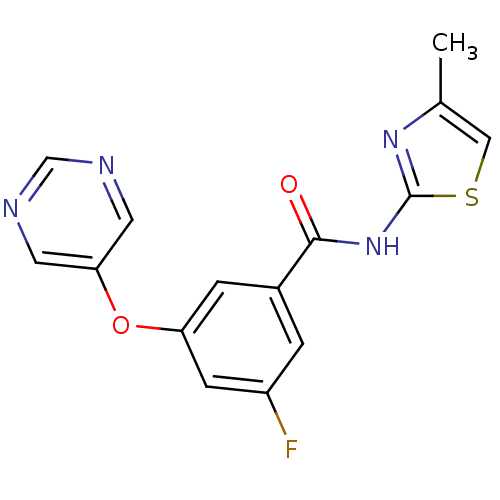
Affinity DataKi: 0.0631nMAssay Description:Effect on pancreatic polypeptide-mediated displacement of 125I-pancreatic polypeptide from human C-terminal eYFP-tagged NY4 receptor expressed in HEK...More data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M4(RAT)
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
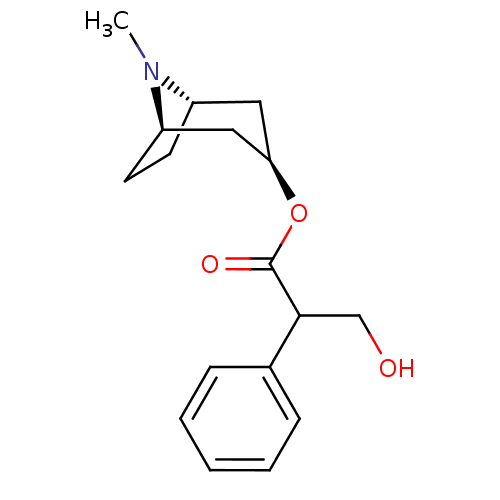
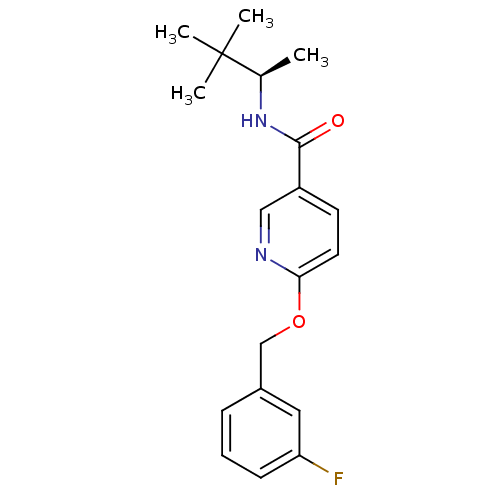
Affinity DataKi: 0.560nMAssay Description:Displacement of [3H]NMS from rat muscarinic M4 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M5(RAT)
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
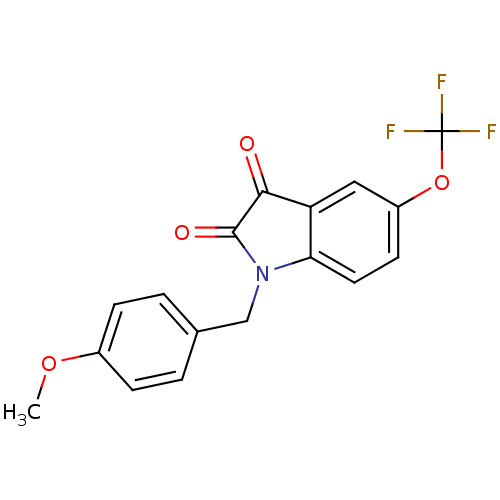
Affinity DataKi: 1.80nMAssay Description:Displacement of [3H]NMS from rat muscarinic M5 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
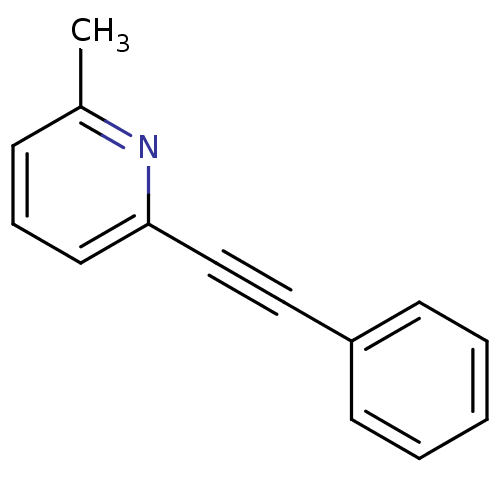
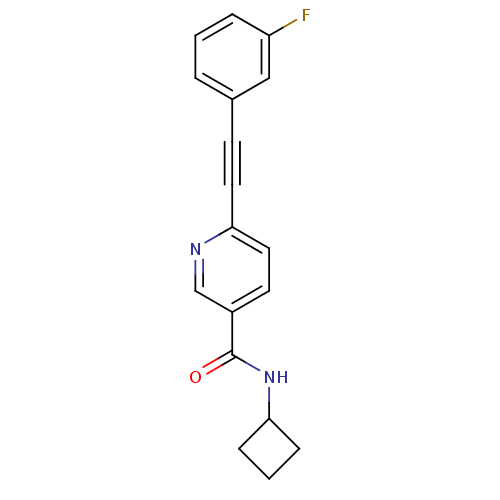
Affinity DataKi: 4.70nMAssay Description:Displacement of [3H]3methoxy-5-(pyridin-2-ylethynyl)pyridine from rat mGluR5More data for this Ligand-Target Pair
Affinity DataKi: 6.30nMAssay Description:Effect on peptide YY-mediated displacement of 125I-pancreatic polypeptide from human C-terminal eYFP-tagged NY4 receptor expressed in HEK293 cell mem...More data for this Ligand-Target Pair
Affinity DataKi: 6.30nMAssay Description:Effect on neuropeptide Y-mediated displacement of 125I-pancreatic polypeptide from human C-terminal eYFP-tagged NY4 receptor expressed in HEK293 cell...More data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M1(RAT)
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Affinity DataKi: 12.7nMAssay Description:Displacement of [3H]NMS from rat muscarinic M1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M3(RAT)
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Affinity DataKi: 74.8nMAssay Description:Displacement of [3H]NMS from rat muscarinic M3 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M5(RAT)
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Affinity DataKi: 85.7nMAssay Description:Displacement of [3H]NMS from rat muscarinic M5 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 116nMAssay Description:Displacement of [3H]3methoxy-5-(pyridin-2-ylethynyl)pyridine from rat mGluR5More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 153nMAssay Description:Displacement of [3H]methoxyPEPy from rat mGluR5 expressed in human HEK293 cells after 1 hr by scintillation counterMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 159nMAssay Description:Displacement of [3H]NMS from human muscarinic M5 receptor expressed in CHO cells coexpressing Gq protein by scintillation counting in presence of ace...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 195nMAssay Description:Displacement of [3H]methoxyPEPy from rat mGluR5 expressed in human HEK293 cells after 1 hr by scintillation counterMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 249nMAssay Description:Displacement of [3H]3methoxy-5-(pyridin-2-ylethynyl)pyridine from rat mGluR5More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 301nMAssay Description:Displacement of [3H]3-methoxy-5-(2-pyridinylethynyl) pyridine from mGluR5More data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M2(RAT)
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Affinity DataKi: 338nMAssay Description:Displacement of [3H]NMS from rat muscarinic M2 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 410nMAssay Description:Displacement of [3H]3-methoxy-5-(2-pyridinylethynyl) pyridine from mGluR5 allosteric siteMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 440nMAssay Description:Negative allosteric modulation at rat mGluR5 receptorMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 440nMAssay Description:Displacement of [3H]3-methoxy-5-(2-pyridinylethynyl) pyridine from mGluR5 allosteric siteMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M4(RAT)
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Vanderbilt Institute Of Chemical Biology
Curated by ChEMBL
Affinity DataKi: 445nMAssay Description:Displacement of [3H]NMS from rat muscarinic M4 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 736nMAssay Description:Displacement of [3H]3methoxy-5-(pyridin-2-ylethynyl)pyridine from rat mGluR5More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 840nMAssay Description:Displacement of [3H]3-methoxy-5-(2-pyridinylethynyl) pyridine from mGluR5 allosteric siteMore data for this Ligand-Target Pair
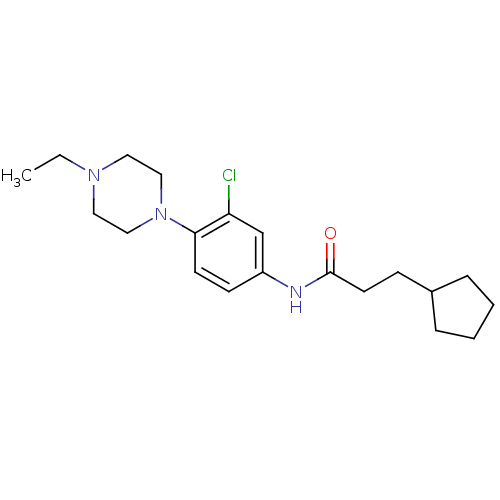
Affinity DataKi: 1.10E+3nMAssay Description:Inhibition of alpha1A adrenergic receptor (unknown origin)More data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: 1.61E+3nMAssay Description:Displacement of [3H]NMS from human muscarinic M5 receptor expressed in CHO cells coexpressing Gq protein by scintillation countingMore data for this Ligand-Target Pair
TargetAlpha-1A/Alpha-1B/Alpha-1D adrenergic receptor(Homo sapiens (Human))
Marquette University
Curated by ChEMBL
Marquette University
Curated by ChEMBL
Affinity DataKi: 1.80E+3nMAssay Description:Inhibition of alpha1 adrenergic receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.50E+3nMAssay Description:Inhibition of PBR receptor (unknown origin)More data for this Ligand-Target Pair
TargetAlpha-1A/Alpha-1B/Alpha-1D adrenergic receptor(Homo sapiens (Human))
Marquette University
Curated by ChEMBL
Marquette University
Curated by ChEMBL
Affinity DataKi: 3.90E+3nMAssay Description:Inhibition of alpha1 adrenergic receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Inhibition of D3 receptor (unknown origin)More data for this Ligand-Target Pair
TargetAlpha-1A/Alpha-1B/Alpha-1D adrenergic receptor(Homo sapiens (Human))
Marquette University
Curated by ChEMBL
Marquette University
Curated by ChEMBL
Affinity DataKi: >1.00E+4nMAssay Description:Inhibition of alpha1 adrenergic receptor (unknown origin)More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataKi: >1.00E+5nMAssay Description:Displacement of [3H]methoxyPEPy from rat mGluR5 expressed in human HEK293 cells after 1 hr by scintillation counterMore data for this Ligand-Target Pair
TargetSmall conductance calcium-activated potassium channel protein 3(Homo sapiens (Human))
Bristol-Myers Squibb
Curated by ChEMBL
Bristol-Myers Squibb
Curated by ChEMBL
Affinity DataIC50: 0.0640nMAssay Description:Inhibition of Kca2.3 channel expressed in HEK293 cells by electrophysiology assayMore data for this Ligand-Target Pair
TargetSmall conductance calcium-activated potassium channel protein 3(Homo sapiens (Human))
Bristol-Myers Squibb
Curated by ChEMBL
Bristol-Myers Squibb
Curated by ChEMBL
Affinity DataIC50: 0.0640nMAssay Description:Inhibition of Kca2.3 channel (unknown origin) expressed in HEK293 cells by electrophysiology assayMore data for this Ligand-Target Pair
TargetSmall conductance calcium-activated potassium channel protein 3(Homo sapiens (Human))
Bristol-Myers Squibb
Curated by ChEMBL
Bristol-Myers Squibb
Curated by ChEMBL
Affinity DataIC50: 0.168nMAssay Description:Inhibition of Kca2.3 channel (unknown origin) expressed in HEK293 cells by thallium flux assayMore data for this Ligand-Target Pair
TargetSmall conductance calcium-activated potassium channel protein 3(Homo sapiens (Human))
Bristol-Myers Squibb
Curated by ChEMBL
Bristol-Myers Squibb
Curated by ChEMBL
Affinity DataIC50: 0.168nMAssay Description:Inhibition of Kca2.3 channel expressed in HEK293 cells by thallium flux assayMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Antagonist activity at mGluR5More data for this Ligand-Target Pair
TargetSmall conductance calcium-activated potassium channel protein 1(Homo sapiens (Human))
Bristol-Myers Squibb
Curated by ChEMBL
Bristol-Myers Squibb
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibition of Kca2.1 channel expressed in HEK293 cells by thallium flux assayMore data for this Ligand-Target Pair
TargetSmall conductance calcium-activated potassium channel protein 3(Homo sapiens (Human))
Bristol-Myers Squibb
Curated by ChEMBL
Bristol-Myers Squibb
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Displacement of [125I]apamin from Kca2.3 channel expressed in HEK293 cells by scintillation proximity assayMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 6.90nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 8.10nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 9.30nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetSmall conductance calcium-activated potassium channel protein 2(Homo sapiens (Human))
Bristol-Myers Squibb
Curated by ChEMBL
Bristol-Myers Squibb
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of Kca2.2 channel expressed in HEK293 cells by thallium flux assayMore data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 15nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 16nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 17nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 19nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 21nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Rattus norvegicus (Rat))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 21nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair
TargetMetabotropic glutamate receptor 5(Homo sapiens (Human))
Vanderbilt University Medical Center
Curated by ChEMBL
Vanderbilt University Medical Center
Curated by ChEMBL
Affinity DataIC50: 23nMAssay Description:The utility of the compounds in accordance with the present invention as negative allosteric modulators of metabotropic glutamate receptor activity, ...More data for this Ligand-Target Pair