Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB (change energy unit to kcal/mol)
 Found 842 hits in this display
Found 842 hits in this display
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Ki: 25nM ΔG°: -43.4kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 7 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration assa...More data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)Ki: 25nM ΔG°: -43.4kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 7 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration assa...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)Ki: 55nM ΔG°: -41.4kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 12 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration ass...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Ki: 146nM ΔG°: -39.0kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 12 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration ass...More data for this Ligand-Target Pair
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)Ki: 568nM ΔG°: -35.6kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 4 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration assa...More data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Arginase(Leishmania amazonensis)Universidade Federal De S£O Carlos
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Ki: 2.40E+3nMAssay Description:Inhibition of MRP1 transfected in human HeLa cells assessed as inhibition of [3H]LTC4 transport by rapid filtration assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Ki: 2.40E+3nMAssay Description:Inhibition of MRP1 transfected in human HeLa cells assessed as inhibition of [3H]LTC4 transport by rapid filtration assayMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)Ki: 2.40E+3nMAssay Description:Inhibition of MRP1 transfected in human HeLa cells assessed as inhibition of [3H]LTC4 transport by rapid filtration assayMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Ki: 3.60E+3nMpH: 7.4Assay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion over a ...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)Ki: 5.60E+3nMAssay Description:Binding affinity to PPARgamma (unknown origin) assessed as inhibition constant by Lanthascreen TR-FRET assay relative to controlMore data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Cholinesterase(Homo sapiens (Human))Institute For Medical Research And Occupational Health
Curated by ChEMBL
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Ki: 9.10E+3nMpH: 7.4Assay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion over a ...More data for this Ligand-Target Pair
Urease subunit beta(Helicobacter pylori (strain ATCC 700392 / 26695) (...)Mazandaran University Of Medical Sciences
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Ki: 9.34E+3nMAssay Description:Inhibition of Helicobacter pylori urease assessed as measuring ammonia production incubated for 1.5 hr by indophenol methodMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Ki: 9.53E+3nM ΔG°: -28.7kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 2 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration assa...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)Ki: >1.00E+4nM ΔG°: >-28.5kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 2 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration assa...More data for this Ligand-Target Pair
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Ki: >1.00E+4nM ΔG°: >-28.5kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 1 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration assa...More data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)Ki: >1.00E+4nM ΔG°: >-28.5kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 1 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration assa...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Ki: >1.00E+4nM ΔG°: >-28.5kJ/moleT: 2°CAssay Description:Inhibition of human recombinant carbonic anhydrase 4 preincubated for 15 mins at room temperature/6 hrs at 4 deg C by stopped-flow CO2 hydration assa...More data for this Ligand-Target Pair
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Ki: 1.31E+4nMAssay Description:Non-competitive inhibition of trypsin (unknown origin) using BApNA as substrate by Lineweaver-Burk/Dixon plot analysisMore data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Ki: 1.38E+4nMpH: 7.4Assay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion over a ...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)Ki: 1.67E+4nMAssay Description:Binding affinity for HA-tagged wild type human Adenosine A2A receptor (WT) using [3H]CGS-21680 as radioligand expressed in COS-7 cellsMore data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)Ki: 1.81E+4nMAssay Description:Binding affinity for HA-tagged mutant human Adenosine A2A receptor (V84L), using [3H]CGS-21680 as radioligand expressed in COS-7 cellsMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)Ki: 3.30E+4nMAssay Description:Binding affinity for HA-tagged mutant human Adenosine A2A receptor (H250N) using [3H]-CGS-21,680 as radioligand expressed in COS-7 cellsMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Cholinesterase(Homo sapiens (Human))Institute For Medical Research And Occupational Health
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Cholinesterase(Homo sapiens (Human))Institute For Medical Research And Occupational Health
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Cholinesterase(Homo sapiens (Human))Institute For Medical Research And Occupational Health
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
Cholinesterase(Homo sapiens (Human))Institute For Medical Research And Occupational Health
Curated by ChEMBL
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Ki: 1.05E+5nM ΔG°: -22.7kJ/molepH: 7.4 T: 2°CAssay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion using s...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Ki: 1.90E+5nMAssay Description:The inhibitor screening assays were performed in 96-well format with 1152 small molecules found in the Prestwick chemical library (Prestwick Chemical...More data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
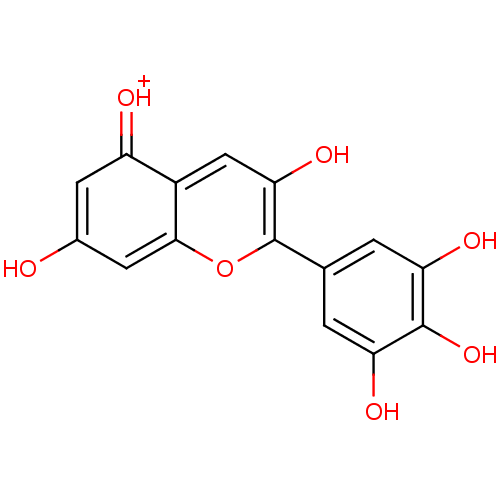 BDBM50326997(CHEMBL590878 | Delphinidin)
BDBM50326997(CHEMBL590878 | Delphinidin)
 BDBM50326997(CHEMBL590878 | Delphinidin)
BDBM50326997(CHEMBL590878 | Delphinidin)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
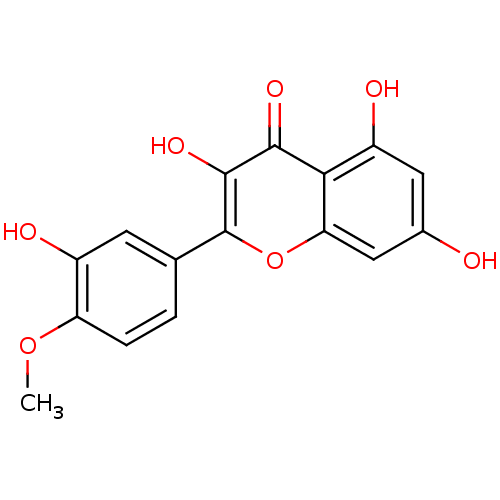 BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM50242286(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3,6-dimethox...)
BDBM50242286(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3,6-dimethox...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
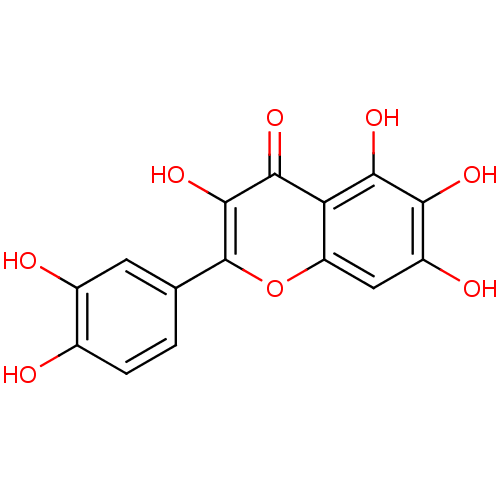 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 220nMAssay Description:Inhibition of alpha-synuclein fibril formation (unknown origin) incubated for 24 hrs to 7 days by thioflavin S based fluorescence assayMore data for this Ligand-Target Pair
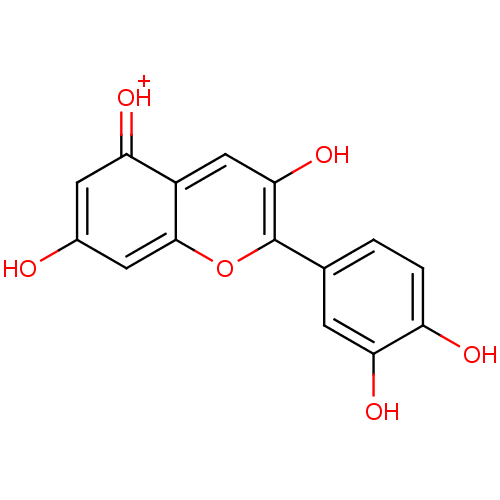 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 350nMAssay Description:Inhibition of human recombinant CK2 expressed in Escherichia coli using RRRADDSDDDDD as substrate after 10 mins in presence of [gamma-32P]ATPMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 360nMAssay Description:Inhibition of human recombinant 17beta-HSD2 expressed in HEK293 cell lysate assessed as conversion of radiolabeled estrone to estradiolMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 400nMAssay Description:Iin vitro inhibition of 5-lipoxygenase activity in rat basophil leukemia type 1(RBL1) cell homogenates, (reduction of [14C]-5-HETE formation)More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 400nMAssay Description:Inhibition of human recombinant CK2 expressed in Escherichia coli using RRRADDSDDDDD as substrate after 10 mins in presence of [gamma-32P]ATPMore data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)IC50: 430nMpH: 7.0 T: 2°CAssay Description:The 96-well flat-bottomed plates were coated with recombinant GST-BAD. After the plates were blocked, the reaction buffer containing test compound an...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)IC50: 500nMAssay Description:Inhibition of human IP6K2 using insP6 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 550nMAssay Description:Inhibition of human recombinant CK2 expressed in Escherichia coli using RRRADDSDDDDD as substrate after 10 mins in presence of [gamma-32P]ATPMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 700nMAssay Description:Inhibition of human IP6K2 using insP6 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 700nMAssay Description:Inhibition of human IP6K2 using insP6 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
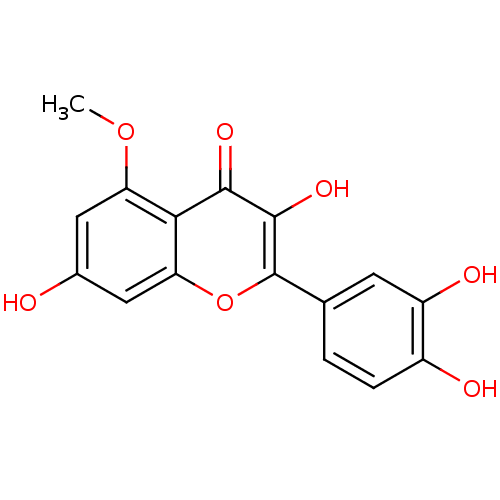 BDBM50326483(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-5-methoxy-4H...)
BDBM50326483(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-5-methoxy-4H...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 780nMpH: 7.0 T: 2°CAssay Description:The 96-well flat-bottomed plates were coated with recombinant GST-BAD. After the plates were blocked, the reaction buffer containing test compound an...More data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 790nMpH: 7.0 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)Sialidase(Clostridium perfringens)Korea Research Institute Of Bioscience And Biotechnology
Curated by ChEMBL
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)Protein E6(Human papillomavirus type 16)Loma Linda University School Of Medicine
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 850nMpH: 7.0 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 850nMpH: 7.0 T: 2°CAssay Description:The 96-well flat-bottomed plates were coated with recombinant GST-BAD. After the plates were blocked, the reaction buffer containing test compound an...More data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 900nMAssay Description:Inhibition of human recombinant CK2 expressed in Escherichia coli using RRRADDSDDDDD as substrate after 10 mins in presence of [gamma-32P]ATPMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.00E+3nMAssay Description:Inhibition of human CYP1B1 expressed in yeast microsomal membranes using 7-ethoxyresorufin as substrate by fluorescence assayMore data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.00E+3nMAssay Description:Inhibition of tyrosinase (unknown origin) using L-tyrosine as substrate assessed as oxidation of L-tyrosine after 15 minsMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.05E+3nMAssay Description:Inhibition of human recombinant 17beta-HSD1 expressed in HEK293 cell lysate assessed as conversion of radiolabeled estrone to estradiolMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 1.10E+3nMAssay Description:Inhibition of human IPMK using insP3 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.10E+3nMpH: 7.0 T: 2°CAssay Description:The 96-well flat-bottomed plates were coated with recombinant GST-BAD. After the plates were blocked, the reaction buffer containing test compound an...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
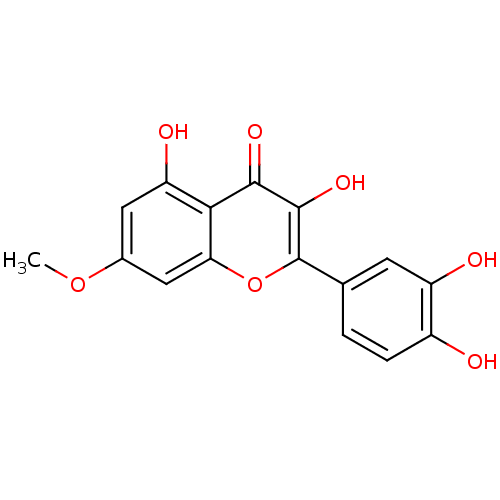 BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)IC50: 1.20E+3nMAssay Description:Inhibition of ELAV3 (unknown origin)-artificial ARE complex formation after 30 mins in the presence of biotin-labeled RNA probe by chemiluminescence ...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.27E+3nMAssay Description:Inhibition of MPO in human neutrophils using H2O2 as substrate measured after 1 hr by fluorescence based HPLC analysisMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 1.30E+3nMAssay Description:Inhibition of Zika virus Asian/8375 NS2B (48 to 100 residues)-NS3 (14 to 185 residues) expressed in Escherichia coli BL21 (DE3) Star cells preincubat...More data for this Ligand-Target Pair
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.30E+3nMpH: 7.0 T: 2°CAssay Description:The 96-well flat-bottomed plates were coated with recombinant GST-BAD. After the plates were blocked, the reaction buffer containing test compound an...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Sialidase(Clostridium perfringens)Korea Research Institute Of Bioscience And Biotechnology
Curated by ChEMBL
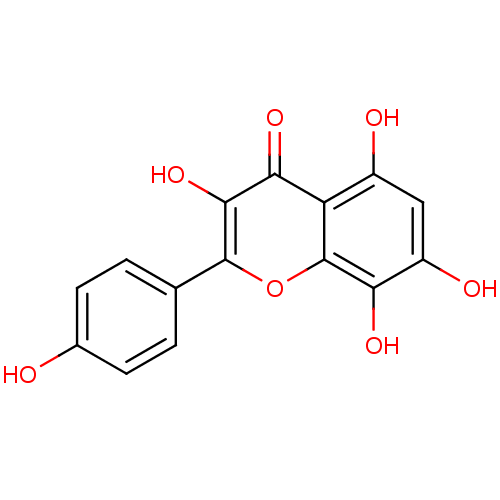 BDBM50304350(CHEMBL611029 | Herbacetin)
BDBM50304350(CHEMBL611029 | Herbacetin)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.40E+3nMAssay Description:Inhibition of ELAV1 (unknown origin)-ARE TNFalpha complex formation after 20 mins by liquid scintillation counting methodMore data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.49E+3nMpH: 7.5Assay Description:The activity of cd-MMP-1 was measured using a fluorescence-based assay. It was performed in white 96-well half area microplate (Greiner) in a final v...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.50E+3nMAssay Description:Inhibition of human thrombin amidolytic activity using D-Phe-Pip-Arg-pNA as substrate preincubated for 10 mins followed by substrate addition measure...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.54E+3nMAssay Description:Inhibition of human recombinant 17beta-HSD2 expressed in HEK293 cell lysate assessed as conversion of radiolabeled estrone to estradiolMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.54E+3nMAssay Description:Inhibition of human 17beta-HSD2 expressed in HEK293 cell lysates incubated for 10 mins using [2,4,6,7-3H]-estradiol and NAD+ by scintillation countin...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
Sialidase(Clostridium perfringens)Korea Research Institute Of Bioscience And Biotechnology
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50554183(CHEBI:75162 | CHEMBL77588)
BDBM50554183(CHEBI:75162 | CHEMBL77588)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)IC50: 1.80E+3nMAssay Description:Inhibition of human IP6K2 using insP6 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.80E+3nMAssay Description:In vitro inhibitory activity against [3H]- (+)-3-PPP binding to Sigma opioid receptor in guinea pig brain membrane homogenatesMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.90E+3nMAssay Description:H-DXR was pre-incubated during 2 min in the presence of the inhibitors at different concentrations and DXP (480 �M). NADPH (160 �M final concentratio...More data for this Ligand-Target Pair
 BDBM50326997(CHEMBL590878 | Delphinidin)
BDBM50326997(CHEMBL590878 | Delphinidin)IC50: 1.90E+3nMAssay Description:Inhibition of human recombinant glyoxalase 1 assessed as S-D-lactoylglutathione after 15 mins by spectrophotometric analysisMore data for this Ligand-Target Pair
 BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)
BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)IC50: 1.90E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Affiliation: Ohio State University Assay Provider: Mam...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.10E+3nMT: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
Arginase(Leishmania amazonensis)Universidade Federal De S£O Carlos
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 2.12E+3nMAssay Description:Desensitization of GPR35 receptor in human HT-29 cells assessed as inhibition of zaprinast-induced dynamic mass redistribution after 10 minsMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)IC50: 2.20E+3nMAssay Description:Inhibition of human IPMK using insP3 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 2.24E+3nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.30E+3nMAssay Description:Inhibition of human IPMK using insP3 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.31E+3nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.44E+3nMAssay Description:Inhibition of recombinant human MAO-A assessed as reduction in 4-hydroxyquinoline formation using kynuramine as substrate after 20 mins by fluorometr...More data for this Ligand-Target Pair
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 2.50E+3nMAssay Description:Inhibition of human IP6K2 using insP6 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 2.60E+3nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 2.63E+3nMAssay Description:The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The...More data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 2.70E+3nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
Sialidase(Clostridium perfringens)Korea Research Institute Of Bioscience And Biotechnology
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)IC50: 2.70E+3nMAssay Description:Inhibition of human recombinant aldose reductase using D-glyceraldehyde as substrate preincubated for 10 mins before substrate addition measured for ...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 2.80E+3nMAssay Description:Inhibition of BACE1 (unknown origin) using Rh-EVNNLDAEFK fluorogenic peptide as substrate incubated for 3 hrs by FRET assayMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.90E+3nMAssay Description:Inhibition of human recombinant aldose reductase using DL-glyceraldehyde, HRAR and beta-NADPH incubated for 10 mins by spectrophotometryMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.92E+3nMAssay Description:Inhibition of DPP4 (unknown origin) using Gly-Pro-AMC as substrate preincubated for 4 secs followed by substrate addition and measured after 30 mins ...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 2.92E+3nMpH: 7.5Assay Description:The activity of cd-MMP-1 was measured using a fluorescence-based assay. It was performed in white 96-well half area microplate (Greiner) in a final v...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM50064891(5,7,3'-trihydroxy-3,6-4'-trimethoxyflavone | 5,7-D...)
BDBM50064891(5,7,3'-trihydroxy-3,6-4'-trimethoxyflavone | 5,7-D...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
PIM2(Homo sapiens (Human))City Of Hope
US Patent
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 3.45E+3nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 3.50E+3nMT: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 3.50E+3nMAssay Description:H-DXR was pre-incubated during 2 min in the presence of the inhibitors at different concentrations and DXP (480 �M). NADPH (160 �M final concentratio...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 3.80E+3nM Kd: 280nMpH: 7.2 T: 2°CAssay Description:Binding was detected as a change in the intrinsic tryptophan fluorescence of the PI3K upon the addition of inhibitor. The inhibitor was incubated wit...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 3.90E+3nMAssay Description:Inhibition of human TRPC5 expressed in HEK293 cells assessed as reduction in gadolinium-induced calcium entry after 30 mins by fluo-4 dye based fluor...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 3.92E+3nMpH: 7.5Assay Description:The activity of cd-MMP-1 was measured using a fluorescence-based assay. It was performed in white 96-well half area microplate (Greiner) in a final v...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 3.96E+3nMAssay Description:The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 3.98E+3nMAssay Description:The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The...More data for this Ligand-Target Pair
 BDBM50240614(3,5-Dihydroxy-2-(3-hydroxy-4-methoxy-phenyl)-7-met...)
BDBM50240614(3,5-Dihydroxy-2-(3-hydroxy-4-methoxy-phenyl)-7-met...)IC50: 3.98E+3nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Arginase(Leishmania amazonensis)Universidade Federal De S£O Carlos
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 4.00E+3nMAssay Description:Inhibition of Leishmania amazonensis recombinant arginase expressed in Escherichia coli Rosetta (DE3) pLysS using L-arginine as substrate incubated f...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 4.03E+3nMAssay Description:The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)IC50: 4.10E+3nMAssay Description:The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The...More data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 4.10E+3nMAssay Description:Inhibition of human amyloid beta (1 to 40) assessed as reduction in aggregation measured after 24 hrs by ThT fluorescence assayMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 4.18E+3nMAssay Description:Inhibition of human recombinant BACE-1 expressed in Escherichia coli using panvera peptide as a substrate incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)IC50: 4.34E+3nMAssay Description:The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 4.40E+3nMAssay Description:Inhibition of human IPMK using insP3 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 4.72E+3nMAssay Description:Inhibition of PI3Kgamma (unknown origin) assessed as decrease in fluorescence intensity using phosphorylated substrateMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Stromelysin-1(Homo sapiens (Human))East China University Of Science And Technology
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
IC50: 6.10E+3nMAssay Description:Inhibition of human microsomal CYP1A2 expressed in baculovirus infected BTI-TN-5B1-4 cells incubated for 30 mins by luciferase assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)IC50: 6.30E+3nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50326997(CHEMBL590878 | Delphinidin)
BDBM50326997(CHEMBL590878 | Delphinidin)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 6.50E+3nMAssay Description:Inhibition of human TRPC5 expressed in HEK293 cells assessed as reduction in gadolinium-induced calcium entry after 30 mins by fluo-4 dye based fluor...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)IC50: 7.39E+3nMAssay Description:The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM50240614(3,5-Dihydroxy-2-(3-hydroxy-4-methoxy-phenyl)-7-met...)
BDBM50240614(3,5-Dihydroxy-2-(3-hydroxy-4-methoxy-phenyl)-7-met...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Neuraminidase(Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...)Prin. K.M. Kundnani College Of Pharmacy
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Sialidase(Clostridium perfringens)Korea Research Institute Of Bioscience And Biotechnology
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Collagenase 3(Homo sapiens (Human))East China University Of Science And Technology
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM50304350(CHEMBL611029 | Herbacetin)
BDBM50304350(CHEMBL611029 | Herbacetin)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)IC50: 9.00E+3nMAssay Description:Inhibition of rabbit skeletal muscle microsomes SERCA1a preincubated for 10 mins followed by addition of ATP and measured after 40 minsMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 9.00E+3nMAssay Description:Inhibition of rabbit skeletal muscle microsomes SERCA1a preincubated for 10 mins followed by addition of ATP and measured after 40 minsMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)IC50: 9.80E+3nMAssay Description:The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The...More data for this Ligand-Target Pair
Sialidase(Clostridium perfringens)Korea Research Institute Of Bioscience And Biotechnology
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 9.90E+3nMAssay Description:Inhibition of self-induced aggregation amyloid beta (1 to 40) (unknown origin) incubated for 24 hrs by Thioflavin T based fluorometric assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
IC50: >1.00E+4nMAssay Description:Inhibition of human microsomal CYP2D6 expressed in baculovirus infected BTI-TN-5B1-4 cells incubated for 30 mins by luciferase assayMore data for this Ligand-Target Pair
IC50: >1.00E+4nMAssay Description:Inhibition of human microsomal CYP2C9 expressed in baculovirus infected BTI-TN-5B1-4 cells incubated for 30 mins by luciferase assayMore data for this Ligand-Target Pair
IC50: >1.00E+4nMAssay Description:Inhibition of human microsomal CYP2C19 expressed in baculovirus infected BTI-TN-5B1-4 cells incubated for 30 mins by luciferase assayMore data for this Ligand-Target Pair
IC50: >1.00E+4nMAssay Description:Inhibition of human microsomal CYP3A4 expressed in baculovirus infected BTI-TN-5B1-4 cells incubated for 30 mins by luciferase assayMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 1.00E+4nMAssay Description:Inhibition of chymotrypsin-like activity of purified human 20S proteasome expressed in human Jurkat cells assessed as decrease in AMC hydrolysis usin...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: >1.00E+4nMAssay Description:Inhibition of human TRPC5 expressed in HEK293 cells assessed as reduction in gadolinium-induced calcium entry after 30 mins by fluo-4 dye based fluor...More data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.00E+4nMAssay Description:Inhibition of human recombinant aldose reductase using D-glyceraldehyde as substrate preincubated for 10 mins before substrate addition measured for ...More data for this Ligand-Target Pair
Sialidase(Clostridium perfringens)Korea Research Institute Of Bioscience And Biotechnology
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.01E+4nMAssay Description:Inhibition of Clostridium perfringens neuraminidase using 4-methylumbelliferyl-alpha-D-N-acetylneuraminic acid as substrate by fluorometryMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.05E+4nMAssay Description:Inhibition of chymotrypsin-like activity of purified human 20S proteasome expressed in human Jurkat cells assessed as decrease in AMC hydrolysis usin...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
Urease subunit beta(Helicobacter pylori (strain ATCC 700392 / 26695) (...)Mazandaran University Of Medical Sciences
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.10E+4nMAssay Description:Inhibition of Helicobacter pylori ATCC 43504 urease-mediated ammonia production preincubated for 1.5 hrs by indophenol methodMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.10E+4nMAssay Description:Inhibition of chymotrypsin-like activity of human 26S proteasome in human Jurkat cells assessed as decrease in AMC hydrolysis using Z-Gly-Gly-Leu-AMC...More data for this Ligand-Target Pair
Urease subunit beta(Helicobacter pylori (strain ATCC 700392 / 26695) (...)Mazandaran University Of Medical Sciences
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Urease subunit beta(Helicobacter pylori (strain ATCC 700392 / 26695) (...)Mazandaran University Of Medical Sciences
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.12E+4nMAssay Description:Inhibition of Helicobacter pylori urease assessed as measuring ammonia production incubated for 1.5 hr by indophenol methodMore data for this Ligand-Target Pair
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)IC50: 1.17E+4nMAssay Description:Inhibition of human recombinant glyoxalase 1 assessed as S-D-lactoylglutathione after 15 mins by spectrophotometric analysisMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 1.20E+4nMAssay Description:Inhibition of chymotrypsin-like activity of human 26S proteasome in human Jurkat cells assessed as decrease in AMC hydrolysis using Z-Gly-Gly-Leu-AMC...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 1.33E+4nMAssay Description:Inhibition of human alpha-synuclein filament formation expressed in Escherichia coli BL21(DE3) cells incubated for 72 hrs by thioflavin S based fluor...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.38E+4nMAssay Description:Inhibition of PI3Kgamma (unknown origin) assessed as decrease in fluorescence intensity using phosphorylated substrateMore data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)IC50: 1.40E+4nMAssay Description:Inhibition of human IPMK using insP3 as substrate preincubated for 15 mins followed by substrate and measured after 30 mins by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.43E+4nMAssay Description:Inhibition of human neutrophil elastase using MeOSuc-AAPV-pNA as substrate measured after 30 mins by spectrometric methodMore data for this Ligand-Target Pair
 BDBM50326997(CHEMBL590878 | Delphinidin)
BDBM50326997(CHEMBL590878 | Delphinidin)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.53E+4nMpH: 7.4 T: 2°CAssay Description:Fluorescence intensity was measured at 420 nm excitation and 485 nm emission using a microplate reader (MPR-A4ιII; TOSOH, Tokyo, Japan, or Fluoroska...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.54E+4nMAssay Description:Desensitization of GPR35 receptor in human HT-29 cells assessed as inhibition of zaprinast-induced dynamic mass redistribution after 10 minsMore data for this Ligand-Target Pair
IC50: 1.63E+4nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.64E+4nMAssay Description:Inhibition of iNOS-mediated nitric oxide production in LPS-stimulated mouse RAW264.7 cells after 24 hrs by Griess reaction analysisMore data for this Ligand-Target Pair
 BDBM84977(6-methoxy kaempferol)
BDBM84977(6-methoxy kaempferol)IC50: 1.72E+4nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.84E+4nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.88E+4nMAssay Description:Inhibition of CYP2D6 in human liver microsomes using dextromethorphan as substrate after 8 mins by LC-MS/MS analysisMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.91E+4nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 1.96E+4nMpH: 6.0 T: 2°CAssay Description:The assay was carried out at room temperature for 10 min with salivary alpha-amylase, starch, and test compounds. The reducing sugar was determined b...More data for this Ligand-Target Pair
 BDBM644029(US11866416, Compound SN-II-92)
BDBM644029(US11866416, Compound SN-II-92)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.00E+4nMpH: 8.0Assay Description:The rate of hydrolysis of the substrates (0.3 mM) by trypsin (18 U/ml), thrombin (12.5 NIH U/ml), urokinase (62500 IU/ml) and elastase (8.4 U/ml) was...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Protein E6(Human papillomavirus type 16)Loma Linda University School Of Medicine
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: >2.00E+4nMAssay Description:Inhibition of GST-tagged Human papillomavirus 16 protein E6 interaction with His-tagged human caspase 8 expressed in Escherichia coli after 1 hr incu...More data for this Ligand-Target Pair
Protein E6(Human papillomavirus type 16)Loma Linda University School Of Medicine
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: >2.00E+4nMAssay Description:Inhibition of GST-tagged Human papillomavirus 16 protein E6 interaction with His-tagged human caspase 8 expressed in Escherichia coli after 1 hr incu...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.00E+4nMAssay Description:Inhibition of human alpha-synuclein filament formation expressed in Escherichia coli BL21(DE3) cells incubated for 72 hrs by thioflavin S based fluor...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)IC50: 2.01E+4nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.14E+4nMpH: 6.0 T: 2°CAssay Description:The assay was carried out at room temperature for 10 min with salivary alpha-amylase, starch, and test compounds. The reducing sugar was determined b...More data for this Ligand-Target Pair
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 2.20E+4nMpH: 7.0 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM84977(6-methoxy kaempferol)
BDBM84977(6-methoxy kaempferol)IC50: 2.24E+4nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.28E+4nMAssay Description:Inhibition of human DNA topoisomerase 2 catalytic domain-mediated knotted bacteriophage P4Virl dell0 DNA unknotting by agarose gel electrophoresisMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.30E+4nMpH: 7.0 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 2.49E+4nMpH: 7.4 T: 2°CAssay Description:The inhibitory activity of flavonoids toward human DPP III was assayed in a 50 mM Tris-HCl buffer, pH 7.4. In brief, recombinant human DPP III (0.29 ...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.50E+4nMpH: 7.0 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
Sialidase(Clostridium perfringens)Korea Research Institute Of Bioscience And Biotechnology
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 2.53E+4nMAssay Description:Inhibition of Clostridium perfringens neuraminidase assessed as inhibition of 4-MU-NANA hydrolysis after 10 mins by spectrofluorometryMore data for this Ligand-Target Pair
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 2.86E+4nMAssay Description:Inhibition of human DNA topoisomerase 2 catalytic domain-mediated knotted bacteriophage P4Virl dell0 DNA unknotting by agarose gel electrophoresisMore data for this Ligand-Target Pair
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
Cruzipain(Trypanosoma cruzi)National Human Genome Research Institute
Curated by ChEMBL
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 3.00E+4nMAssay Description:Inhibition of thrombin (unknown origin) assessed as hydrolysis of N-benzoyl-phenylalanylvalyl-arginine-paranitroanilideMore data for this Ligand-Target Pair
 BDBM50326997(CHEMBL590878 | Delphinidin)
BDBM50326997(CHEMBL590878 | Delphinidin)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 3.02E+4nMpH: 6.0 T: 2°CAssay Description:The assay was carried out at room temperature for 10 min with salivary alpha-amylase, starch, and test compounds. The reducing sugar was determined b...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)
BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 3.16E+4nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 3.19E+4nMAssay Description:Inhibition of bovine milk xanthine oxidase assessed using xanthine as substrate after 10 mins by fluorometric assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 3.24E+4nMpH: 7.45 T: 2°CAssay Description:Inhibition assay of human testes mcrosomal 17 beta-hydroxysteroid dehydrogenase for the reduction of androstenedione.More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 3.29E+4nMpH: 7.4 T: 2°CAssay Description:The inhibitory activity of flavonoids toward human DPP III was assayed in a 50 mM Tris-HCl buffer, pH 7.4. In brief, recombinant human DPP III (0.29 ...More data for this Ligand-Target Pair
 BDBM50304350(CHEMBL611029 | Herbacetin)
BDBM50304350(CHEMBL611029 | Herbacetin)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50064891(5,7,3'-trihydroxy-3,6-4'-trimethoxyflavone | 5,7-D...)
BDBM50064891(5,7,3'-trihydroxy-3,6-4'-trimethoxyflavone | 5,7-D...)
Prothrombin(Bos taurus (Bovine))Slovak University Of Technology
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 3.50E+4nMpH: 8.0Assay Description:The rate of hydrolysis of the substrates (0.3 mM) by trypsin (18 U/ml), thrombin (12.5 NIH U/ml), urokinase (62500 IU/ml) and elastase (8.4 U/ml) was...More data for this Ligand-Target Pair
 BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)
BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50242286(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3,6-dimethox...)
BDBM50242286(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3,6-dimethox...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 3.77E+4nMAssay Description:Inhibition of human DNA topoisomerase 2 catalytic domain-mediated knotted bacteriophage P4Virl dell0 DNA unknotting by agarose gel electrophoresisMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 3.81E+4nMAssay Description:Inhibition of Influenza A Jinan/15/90 H3N2 virus neuraminidase activity by MUN-ANA substrate based fluorimetric assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 3.87E+4nMAssay Description:Inhibition of recombinant human MAO-B assessed as reduction in 4-hydroxyquinoline formation using kynuramine as substrate after 20 mins by fluorometr...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 4.10E+4nMpH: 7.0 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
Neuraminidase(Influenza A virus (strain A/USSR/90/1977 H1N1))Freie Universitaet Berlin
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)
BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)IC50: 4.49E+4nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Affiliation: Ohio State University Assay Provider: Mam...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 4.62E+4nMAssay Description:Inhibition of Influenza A Jinan/15/90 H3N2 virus neuraminidase activity by MUN-ANA substrate based fluorimetric assayMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 4.64E+4nMAssay Description:Inhibition of Influenza A Jiangsu/10/2003 virus neuraminidase activity by MUN-ANA substrate based fluorimetric assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
BDBM23408(2-(3,4-dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 5.00E+4nMpH: 8.0Assay Description:The rate of hydrolysis of the substrates (0.3 mM) by trypsin (18 U/ml), thrombin (12.5 NIH U/ml), urokinase (62500 IU/ml) and elastase (8.4 U/ml) was...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 5.10E+4nMpH: 7.0 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Arginase(Leishmania amazonensis)Universidade Federal De S£O Carlos
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 5.50E+4nMAssay Description:Inhibition of Leishmania amazonensis recombinant arginase expressed in Escherichia coli Rosetta (DE3) pLysS using L-arginine as substrate incubated f...More data for this Ligand-Target Pair
 BDBM50240614(3,5-Dihydroxy-2-(3-hydroxy-4-methoxy-phenyl)-7-met...)
BDBM50240614(3,5-Dihydroxy-2-(3-hydroxy-4-methoxy-phenyl)-7-met...)IC50: 5.54E+4nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
Neuraminidase(Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...)Prin. K.M. Kundnani College Of Pharmacy
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Neuraminidase(Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...)Prin. K.M. Kundnani College Of Pharmacy
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
Neuraminidase(Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...)Prin. K.M. Kundnani College Of Pharmacy
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)Neuraminidase(Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...)Prin. K.M. Kundnani College Of Pharmacy
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Cruzipain(Trypanosoma cruzi)National Human Genome Research Institute
Curated by ChEMBL
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
IC50: 6.09E+4nMT: 2°CAssay Description:The p38alpha reaction was carried out by using kinase (12ng per well), ATP (100uM) and incubated for 60 min at 37 C. For the JNK3 assay, kinase (10n...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 6.75E+4nMAssay Description:Inhibition of Influenza A Jiangsu/10/2003 virus neuraminidase activity by MUN-ANA substrate based fluorimetric assayMore data for this Ligand-Target Pair
 BDBM50064891(5,7,3'-trihydroxy-3,6-4'-trimethoxyflavone | 5,7-D...)
BDBM50064891(5,7,3'-trihydroxy-3,6-4'-trimethoxyflavone | 5,7-D...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: >7.00E+4nMAssay Description:Inhibition of porcine pancreatic lipase using p-nitrophenylbutyrate as substrate assessed as formation of p-nitrophenol preincubated for 15 mins foll...More data for this Ligand-Target Pair
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)IC50: >7.00E+4nMAssay Description:Inhibition of porcine pancreatic lipase using p-nitrophenylbutyrate as substrate assessed as formation of p-nitrophenol preincubated for 15 mins foll...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 7.41E+4nMpH: 7.4 T: 2°CAssay Description:The inhibitory activity of flavonoids toward human DPP III was assayed in a 50 mM Tris-HCl buffer, pH 7.4. In brief, recombinant human DPP III (0.29 ...More data for this Ligand-Target Pair
 BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)
BDBM50033767(3,3',4',5',7-pentahydroxy flavone | 3,7,3',4',5'-P...)IC50: 7.45E+4nMAssay Description:A previously reported zymographic assay was utilized for the investigation of Gtf enzymatic activity, as described in Example 1. A 1:100 in fresh 5 m...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 7.50E+4nMpH: 7.0 T: 2°CAssay Description:Kinase activities were assayed in buffers containing substrate, enzyme, and inhibitor at 30 °C in the presence of 15 uM ATP/ [gamma-32P] ATP. 32...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 7.51E+4nMpH: 7.4 T: 2°CAssay Description:Fluorescence intensity was measured at 420 nm excitation and 485 nm emission using a microplate reader (MPR-A4ιII; TOSOH, Tokyo, Japan, or Fluoroska...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 7.54E+4nMAssay Description:Inhibition of Influenza A Jiangsu/10/2003 virus neuraminidase activity by MUN-ANA substrate based fluorimetric assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Neuraminidase(Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...)Prin. K.M. Kundnani College Of Pharmacy
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
Neuraminidase(Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...)Prin. K.M. Kundnani College Of Pharmacy
Curated by ChEMBL
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
Neuraminidase(Influenza A virus (strain A/USSR/90/1977 H1N1))Freie Universitaet Berlin
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 8.76E+4nMAssay Description:Inhibition of Influenza A Jinan/15/90 H3N2 virus neuraminidase activity by MUN-ANA substrate based fluorimetric assayMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)IC50: 9.37E+4nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
Integrase(Human immunodeficiency virus 1)National Cancer Institute-Bethesda
Curated by ChEMBL
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)
BDBM50325674(3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-...)IC50: 1.00E+5nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: >1.00E+5nMAssay Description:Displacement of fluorescent SM5F peptide from XIAP linker BIR3 domain after 3 hrs by fluorescence polarization assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: >1.00E+5nMAssay Description:Inhibition of porcine pancreatic lipase pre-incubated for 15 mins before p-nitrophenylbutyrate substrate addition by microplate reader based methodMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 1.01E+5nMpH: 7.45 T: 2°CAssay Description:Inhibition assay of human testes mcrosomal 17 beta-hydroxysteroid dehydrogenase for the reduction of androstenedione.More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 1.50E+5nMAssay Description:Inhibition of human Neu2 assessed as MuNANA substrate hydrolysis in presence of 0.1% Triton X-100 by discontinuous fluorimetric assayMore data for this Ligand-Target Pair
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.55E+5nMAssay Description:In vitro antibacterial activity was determined as inhibitory concentration causing 50% DNA-gyrase supercoiling inhibition (SCI)More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.55E+5nMAssay Description:In vitro antibacterial activity was determined as inhibitory concentration causing 50% DNA-gyrase supercoiling inhibition (SCI)More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 1.60E+5nMAssay Description:Inhibition of human Neu2 assessed as MuNANA substrate hydrolysis in presence of 0.1% Triton X-100 by discontinuous fluorimetric assayMore data for this Ligand-Target Pair
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: >2.00E+5nMAssay Description:Inhibition of human SET7 overexpressed in Escherichia coli BL21 (DE3) cells preincubated for 15 mins followed by addition of SAM as substrate and bio...More data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 2.30E+5nMAssay Description:Inhibition of human Neu2 assessed as MuNANA substrate hydrolysis in presence of 0.1% Triton X-100 by discontinuous fluorimetric assayMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
BDBM26655(2-(3,4-dihydroxyphenyl)-3,5,7,8-tetrahydroxy-4H-ch...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 2.89E+5nMAssay Description:In vitro antibacterial activity was determined as inhibitory concentration causing 50% DNA-gyrase supercoiling inhibition (SCI)More data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: >3.00E+5nMAssay Description:Inhibition of CMG2 (40 to 217) C175A and R40C double mutant (unknown origin) interaction to full length PA E733C mutant expressed in Escherichia coli...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 3.89E+5nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 3.98E+5nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
Cholinesterase(Homo sapiens (Human))Institute For Medical Research And Occupational Health
Curated by ChEMBL
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)IC50: 4.81E+5nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)IC50: >5.00E+5nMpH: 7.4 T: 2°CAssay Description:Fluorescence intensity was measured at 420 nm excitation and 485 nm emission using a microplate reader (MPR-A4ιII; TOSOH, Tokyo, Japan, or Fluoroska...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)IC50: 5.01E+5nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 7.07E+5nMAssay Description:In vitro antibacterial activity was determined as inhibitory concentration causing 50% DNA-gyrase supercoiling inhibition (SCI)More data for this Ligand-Target Pair
IC50: 1.38E+6nMAssay Description:In vitro antibacterial activity was determined as inhibitory concentration causing 50% DNA-gyrase supercoiling inhibition (SCI)More data for this Ligand-Target Pair
IC50: >1.38E+6nMAssay Description:In vitro antibacterial activity was determined as inhibitory concentration causing 50% DNA-gyrase supercoiling inhibition (SCI)More data for this Ligand-Target Pair
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)IC50: >1.85E+6nMAssay Description:In vitro antibacterial activity was determined as inhibitory concentration causing 50% DNA-gyrase supercoiling inhibition (SCI)More data for this Ligand-Target Pair
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)IC50: 4.00E+9nMAssay Description:Inhibition of baker's yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate by spectrophotometryMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)IC50: 5.00E+9nMAssay Description:Inhibition of baker's yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate by spectrophotometryMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)IC50: 7.00E+9nMAssay Description:Inhibition of baker's yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate by spectrophotometryMore data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)IC50: 1.20E+10nMAssay Description:Inhibition of baker's yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate by spectrophotometryMore data for this Ligand-Target Pair
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)IC50: 1.30E+10nMAssay Description:Inhibition of baker's yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate by spectrophotometryMore data for this Ligand-Target Pair
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)EC50: 3.50E+3nMAssay Description:Agonist activity at human LXRalpha-LBD assessed as recruitment of co-activator peptide after 2 hrs by TR-FRET assayMore data for this Ligand-Target Pair
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)pH: 6.0 T: 2°CAssay Description:The assay was carried out at room temperature for 10 min with salivary alpha-amylase, starch, and test compounds. The reducing sugar was determined b...More data for this Ligand-Target Pair
 BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)
BDBM23409(3,5,7-trihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-...)pH: 6.0 T: 2°CAssay Description:The assay was carried out at room temperature for 10 min with salivary alpha-amylase, starch, and test compounds. The reducing sugar was determined b...More data for this Ligand-Target Pair
 BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)
BDBM23410(2-(3,4-dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H...)pH: 6.0 T: 2°CAssay Description:The assay was carried out at room temperature for 10 min with salivary alpha-amylase, starch, and test compounds. The reducing sugar was determined b...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)EC50: 1.80E+4nMAssay Description:Keywords: STK33 Kinase, Non-ATP Competitive Inhibitor Assay Overview: Purified STK33 Kinase is preincubated with potential inhibitors and allowed to ...More data for this Ligand-Target Pair
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)EC50: 1.16E+4nMAssay Description:Keywords: GSK3beta, dose response, kinase, inhibition, HTS Assay Overview: The glycogen synthase kinase-3 beta (GSK-3b) is a known master regulator f...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Kd: 1.70E+3nMpH: 4.0Assay Description:Recombinant full-length hnRNPA1 (aa 1-320) and truncated versions of hnRNPA1, including the N-terminal RNA binding domain (aa 1-196), the middle regi...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
BDBM50241503(2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-Benzopy...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
BDBM7457(2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
BDBM50049391(3,5,7-Trihydroxyflavone | 3,5,7-triOH-flavone | 3,...)
 BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
BDBM7462(3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)EC50: 1.47E+4nMAssay Description:Agonist activity at GPR35 receptor in human U2OS cells coexpressing Gal4-VP16-TEV assessed as beta arrestin translocation after 5 hrs by beta lactmas...More data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Kd: 2.56E+4nMAssay Description:Binding affinity to sensorchip-immobilized human His-tagged CFTR F508 deletion mutant by surface plasmon resonance analysisMore data for this Ligand-Target Pair
 BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)
BDBM7460(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Kd: 5.18E+4nMAssay Description:Binding affinity to human MMP-1 catalytic domain (100 to 269 residues) expressed in Escherichia coli BL21 Star (DE3) by SPR analysisMore data for this Ligand-Target Pair
 BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)
BDBM50240896(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-methoxy-4H...)Kd: 2.17E+4nMAssay Description:Binding affinity to human MMP-1 catalytic domain (100 to 269 residues) expressed in Escherichia coli BL21 Star (DE3) by SPR analysisMore data for this Ligand-Target Pair
 BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
BDBM15236(3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chr...)
Displayed 1 to 50 (of 842 total ) | Next | Last >>