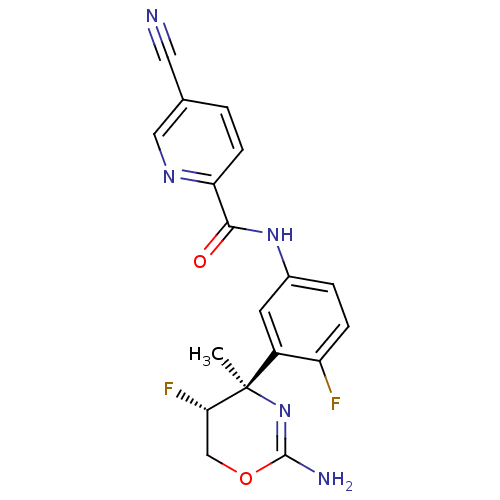
Affinity DataIC50: 1nMT: 2°CAssay Description:The assay uses the principle of inhibition of human TMEM27 cleavage by endogenous cellular BACE2 in the Ins1e rat cell line and shedding from the cel...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human BACE1 in HEK293 cells transfected with wild type APP assessed as reduction of amyloid beta40 level after 18 to 20 hrs by AlphaLIS...More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Inhibition of human BACE1 using MR121-labeled substrate incubated for 4 mins prior to substrate addition measured after 2 mins by spectrophotometric ...More data for this Ligand-Target Pair
Affinity DataIC50: 48nMAssay Description:Inhibition of recombinant human BACE1 (43 to 454 residues) expressed in Escherichia coli BL21(DE3) using Biotin-epsilon-amino-n-capronic acid-SEVNLDA...More data for this Ligand-Target Pair

 3D Structure (crystal)
3D Structure (crystal)