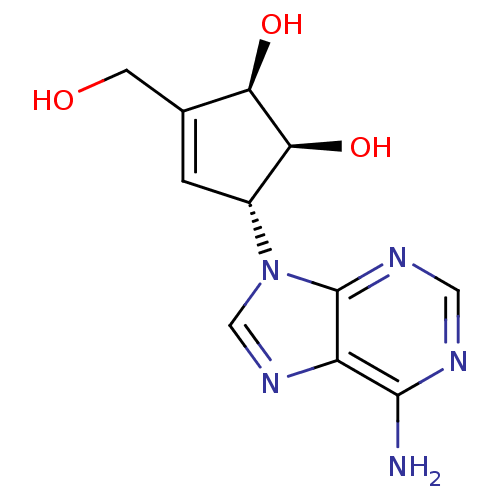
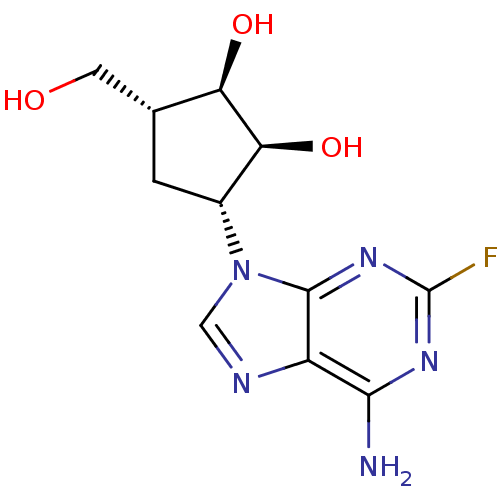
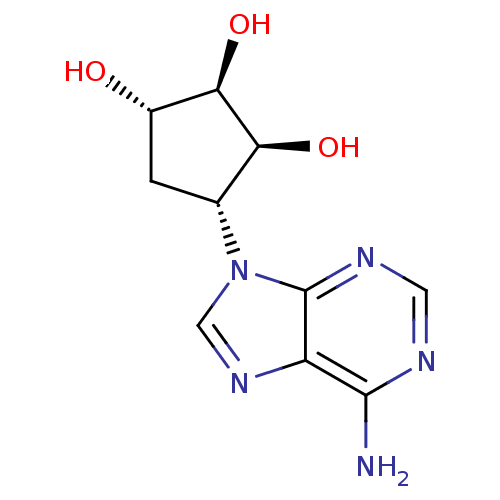
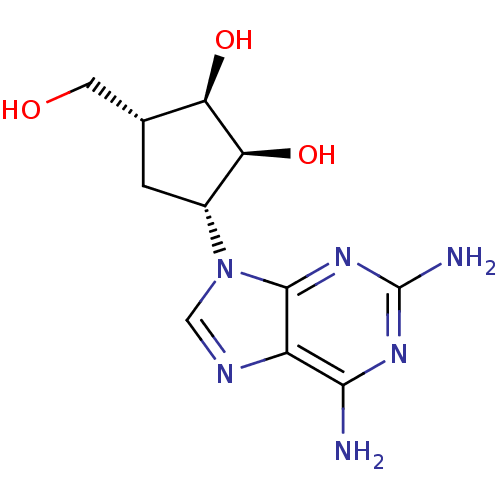
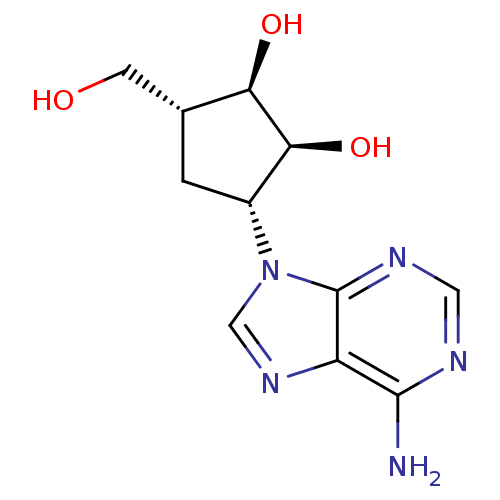
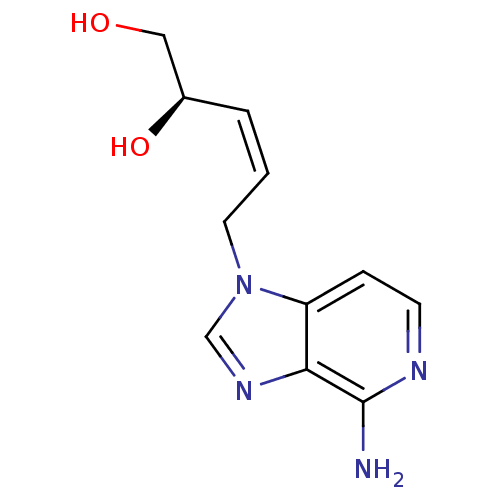
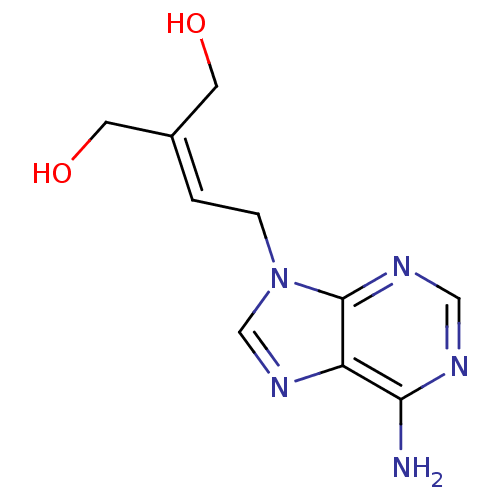
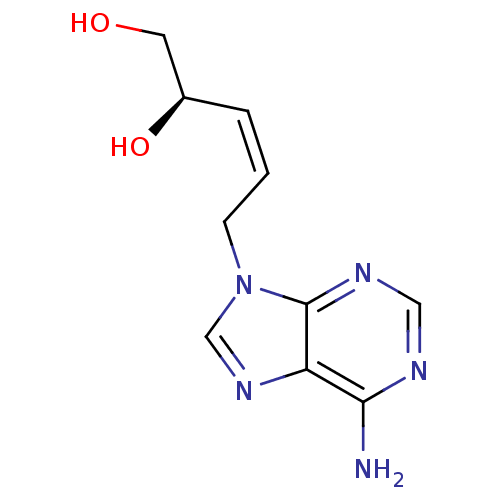
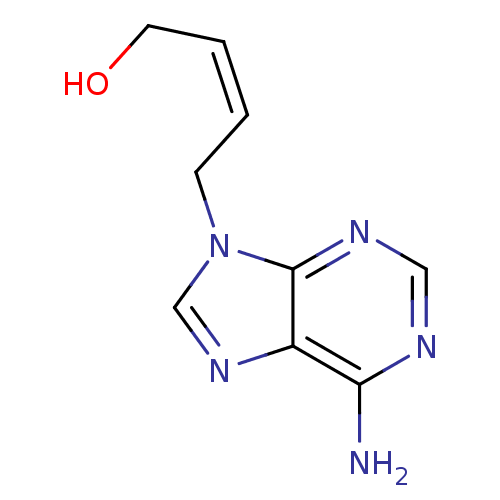
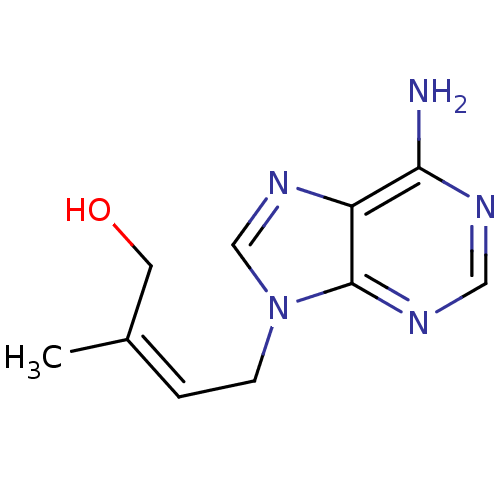
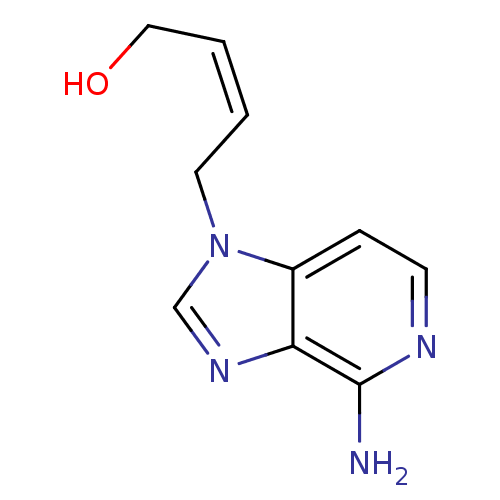
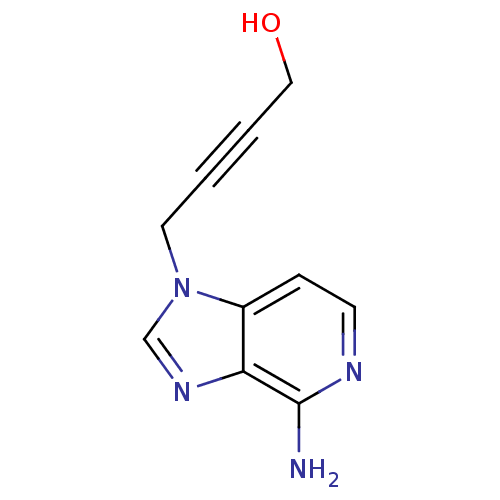
Affinity DataIC50: 50nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.98E+3nMAssay Description:Inhibition of Plasmodium falciparum SAHHMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of Plasmodium falciparum SAHHMore data for this Ligand-Target Pair
Affinity DataIC50: 3.10E+3nMAssay Description:Inhibitory activity of the compound against P. falciparum S-adenosyl-L-homocysteine hydrolaseMore data for this Ligand-Target Pair
Affinity DataIC50: 3.10E+3nMAssay Description:Inhibition of Plasmodium falciparum SAHHMore data for this Ligand-Target Pair
Affinity DataIC50: 4.51E+3nMAssay Description:Inhibition of Plasmodium falciparum SAHHMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMAssay Description:Inhibitory activity of the compound against P. falciparum S-adenosyl-L-homocysteine hydrolaseMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMAssay Description:Inhibition of Plasmodium falciparum SAHHMore data for this Ligand-Target Pair
Affinity DataIC50: 5.70E+4nMAssay Description:Inhibition of Plasmodium falciparum SAHHMore data for this Ligand-Target Pair
Affinity DataIC50: 6.10E+4nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 6.50E+4nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+5nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.45E+5nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+5nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+5nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+5nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+5nMAssay Description:Evaluated for the 50% inhibition of S-Adenosyl-homocysteine (AdoHcy) hydrolase L929 lysate from murine L-929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: >5.00E+5nMAssay Description:In vitro inhibitory concentration of the compound against purified S-Adenosylhomocysteine hydrolase isolated from murine L929 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: >5.00E+5nMAssay Description:In vitro inhibitory concentration of the compound against purified S-Adenosylhomocysteine hydrolase isolated from murine L929 cellsMore data for this Ligand-Target Pair