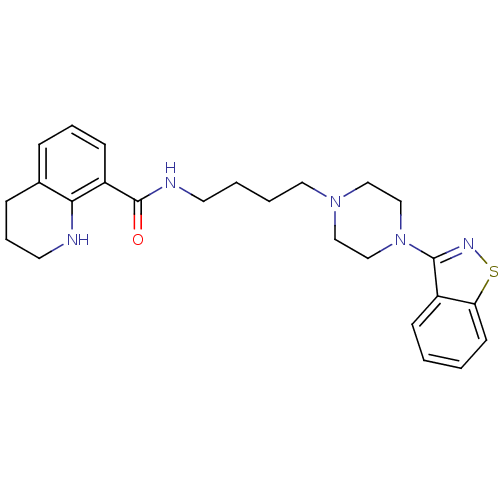
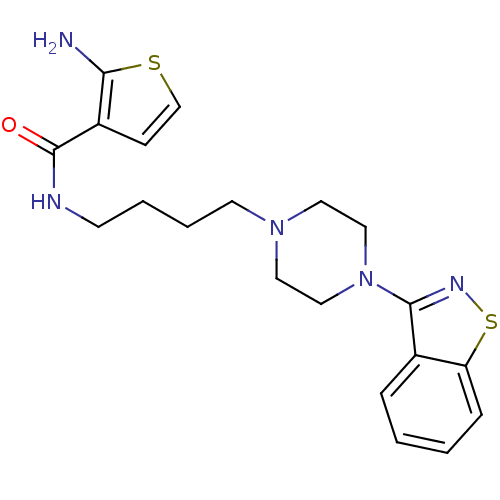
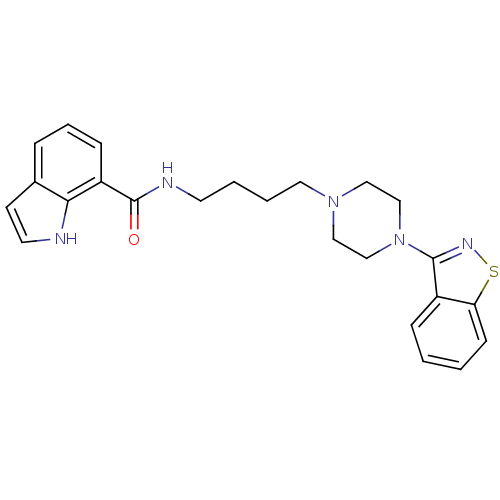
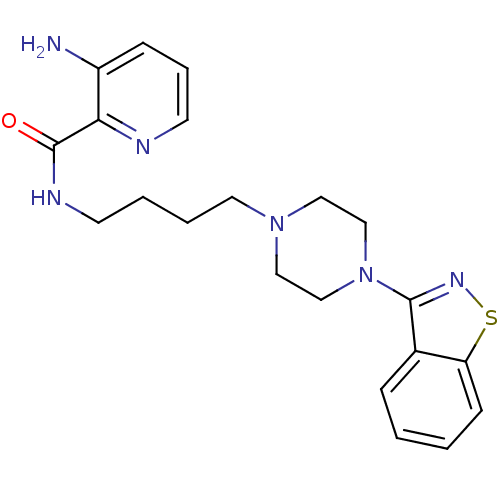
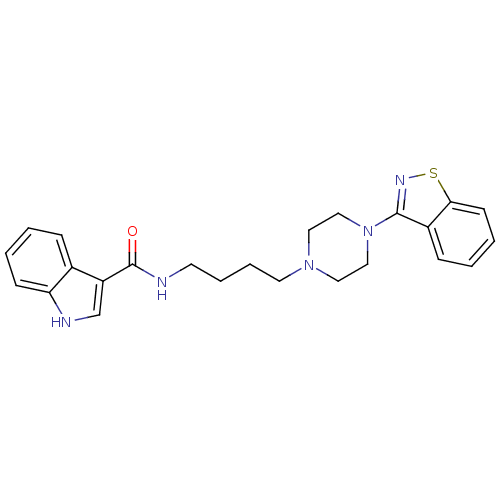
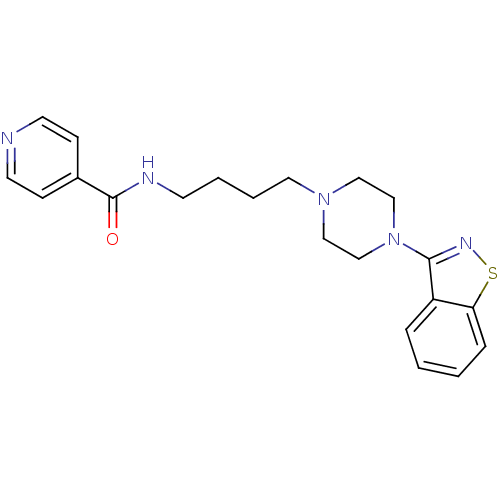
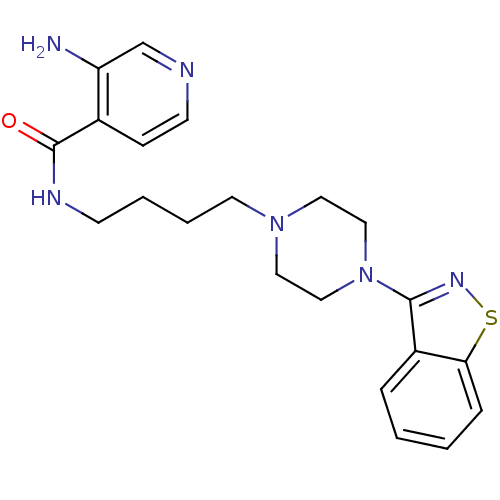
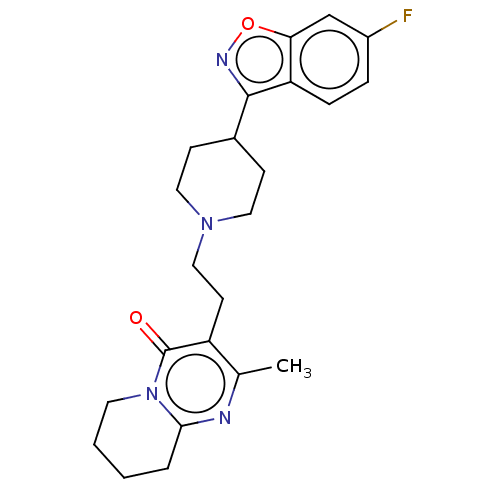
Affinity DataIC50: 1.30nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 2.70nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 2.80nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 3.30nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 3.5nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 3.60nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 3.80nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 4.20nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 4.30nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 4.30nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 4.70nMAssay Description:Inhibitory activity against Dopamine receptor D2 from miceMore data for this Ligand-Target Pair
Affinity DataIC50: 4.90nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 5.10nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 5.80nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 5.80nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 5.80nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 6.60nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 7.60nMAssay Description:Inhibitory activity against Dopamine receptor D2 from miceMore data for this Ligand-Target Pair
Affinity DataIC50: 7.80nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 9.60nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Inhibitory activity against Dopamine receptor D2 from miceMore data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Inhibitory activity against Dopamine receptor D2 from miceMore data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Inhibitory activity against Dopamine receptor D2 from miceMore data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Inhibitory activity against Dopamine receptor D2 from miceMore data for this Ligand-Target Pair
Affinity DataIC50: 23nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:Inhibitory activity against Dopamine receptor D2 from miceMore data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Inhibitory activity against Dopamine receptor D2 from miceMore data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 34nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 47nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 290nMAssay Description:Inhibition of mouse Dopamine receptor D2More data for this Ligand-Target Pair
Affinity DataIC50: 430nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibitory activity against serotonin 5-hydroxytryptamine 1A receptor from mice.More data for this Ligand-Target Pair