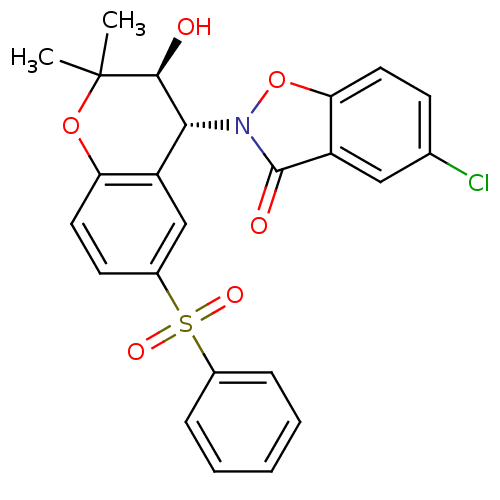
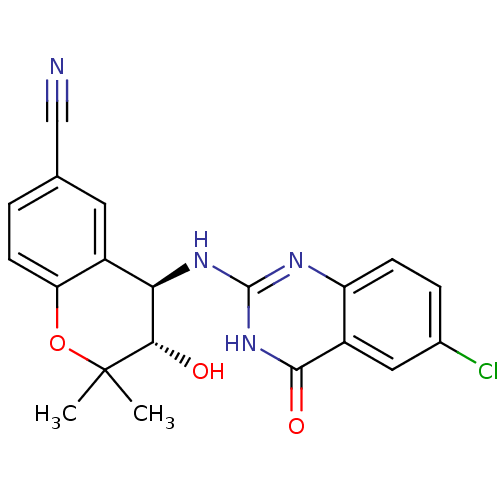
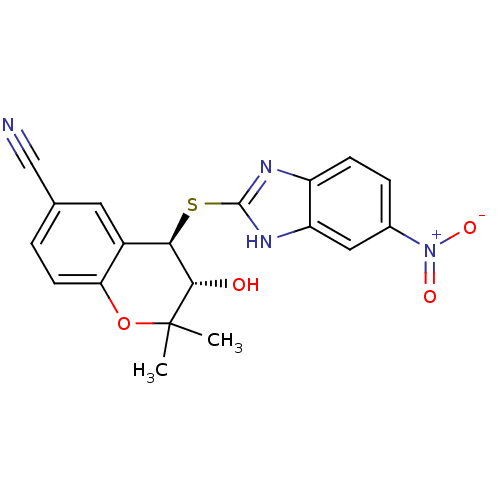
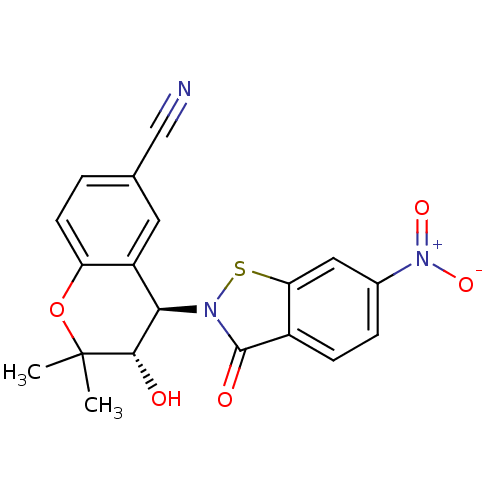
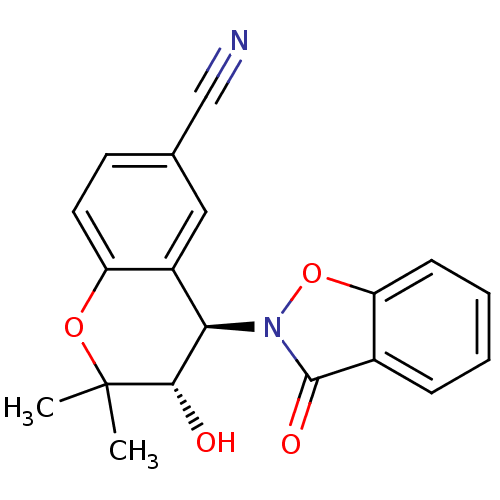
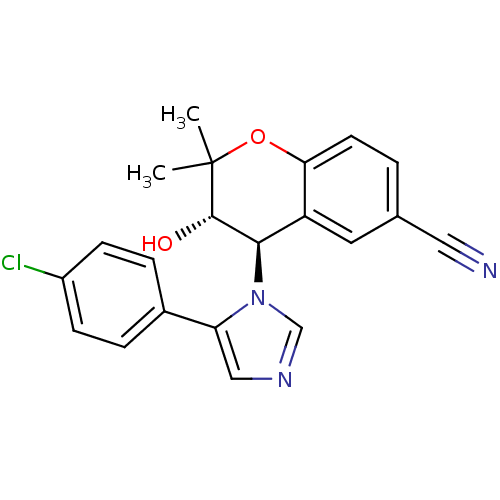
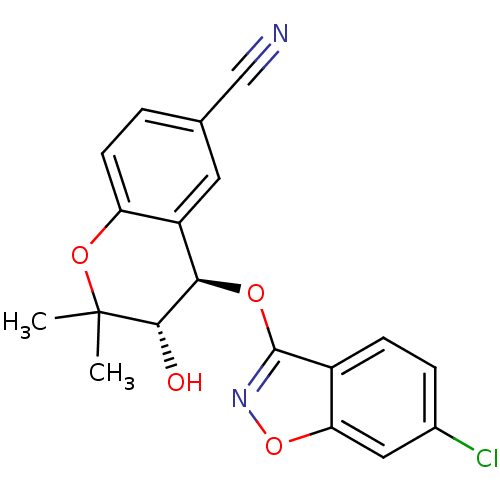
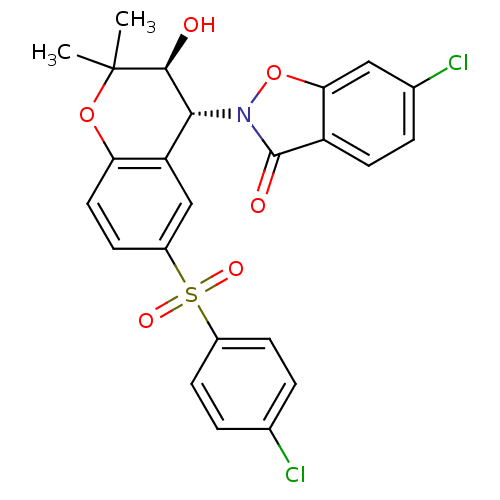
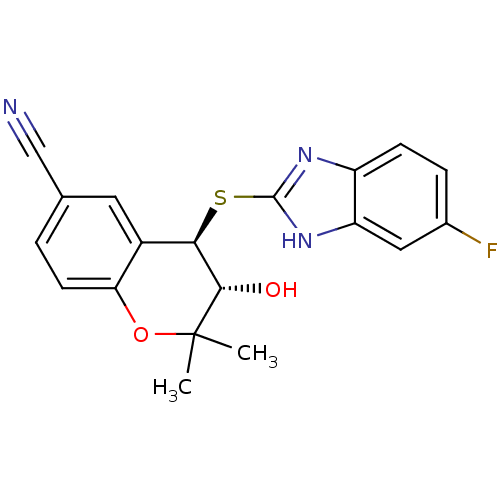
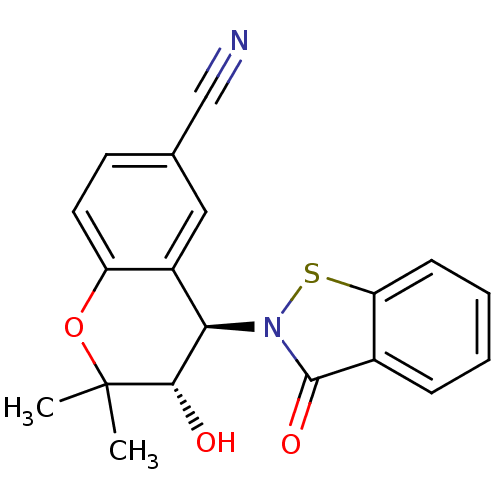
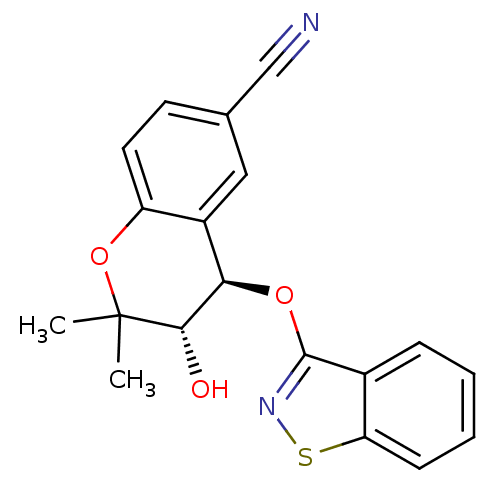
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
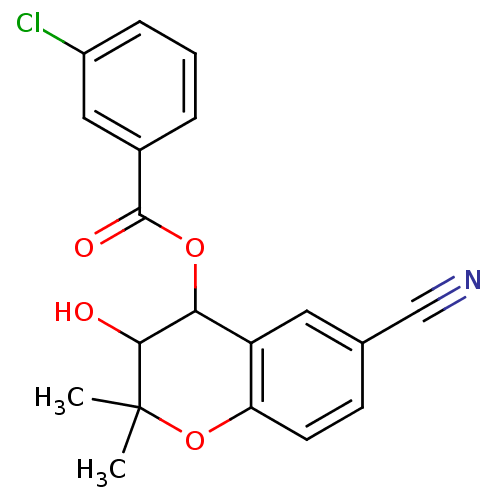
Affinity DataEC50: 70nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
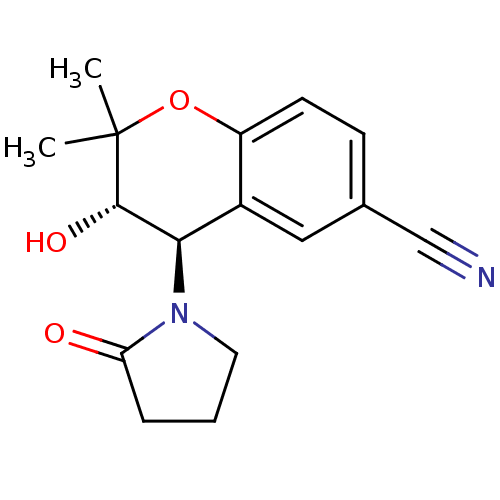
Affinity DataEC50: 120nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 230nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 310nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 560nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 670nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 750nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 780nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 810nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 820nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.08E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.25E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.37E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.50E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.54E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.56E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.64E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.71E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 2.07E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 2.29E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 2.44E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 2.60E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 2.75E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 3.09E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 3.16E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 3.54E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 3.71E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 4.10E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 4.33E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 6.04E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 6.18E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 6.45E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 6.85E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 6.94E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 7.06E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 7.33E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 7.45E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 7.77E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 7.85E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 7.99E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 8.44E+3nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.03E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.29E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.30E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.38E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.41E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.47E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.61E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.65E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 9(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataEC50: 1.86E+4nMAssay Description:Channel opening activity at SUR2B/Kir6.2 potassium ATP channel in human TE671 cells assessed as isometric force by FLIPRMore data for this Ligand-Target Pair










 3D Structure (crystal)
3D Structure (crystal)