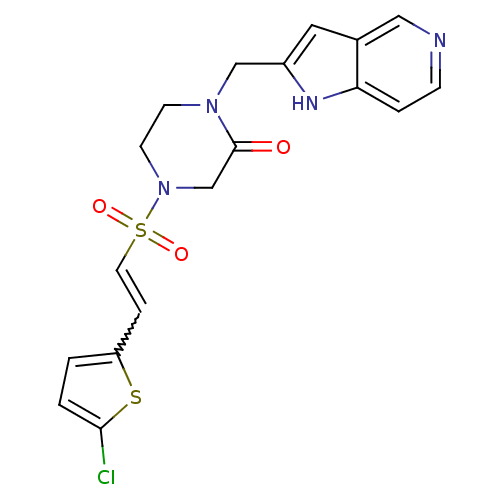
Affinity DataKi: 1.10nM ΔG°: -50.6kJ/molepH: 7.5 T: 2°CAssay Description:The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Inhibition of factor 10aMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Binding affinity (in vitro) of the compound towards human Coagulation factor X was determined at 5 mg/kg peroral doseMore data for this Ligand-Target Pair

 3D Structure (crystal)
3D Structure (crystal)