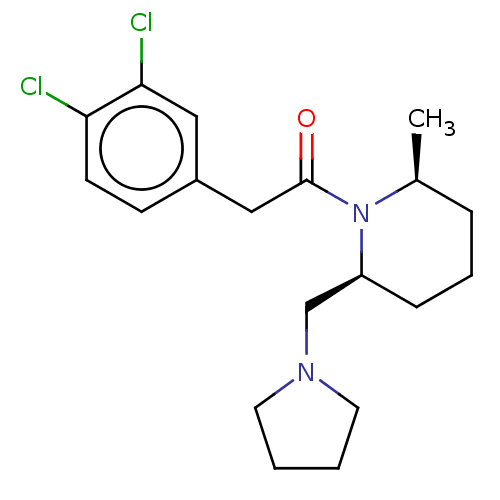
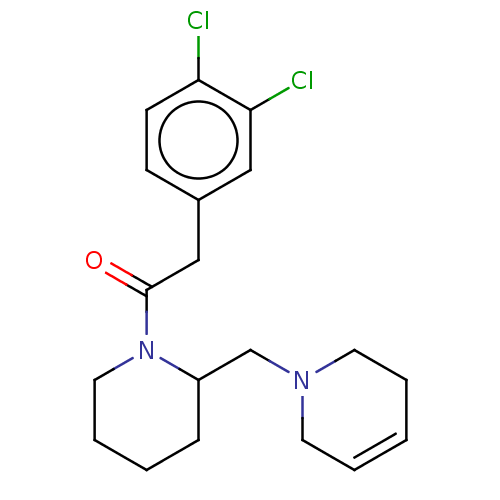
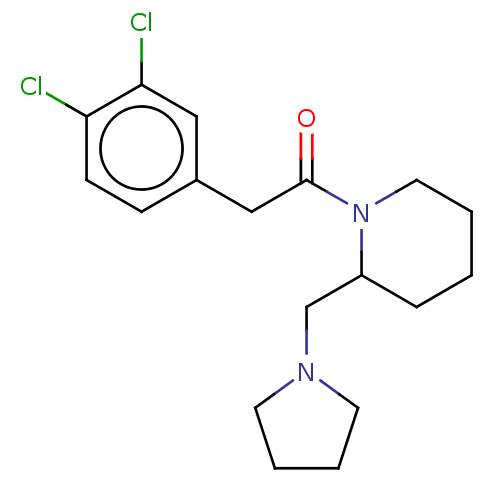
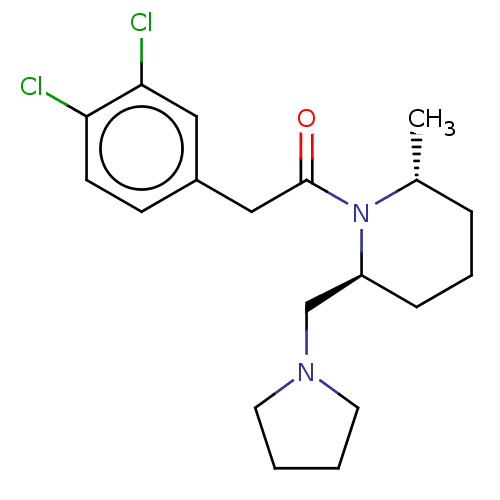
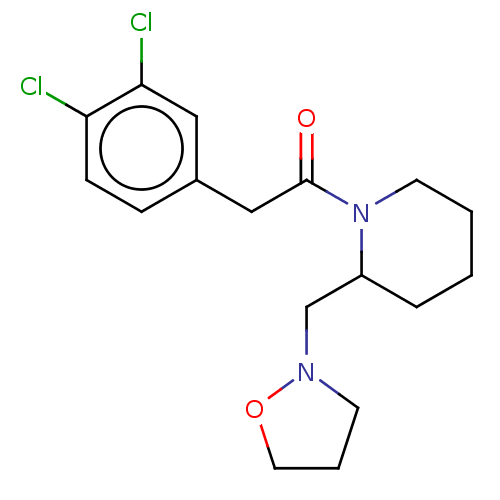
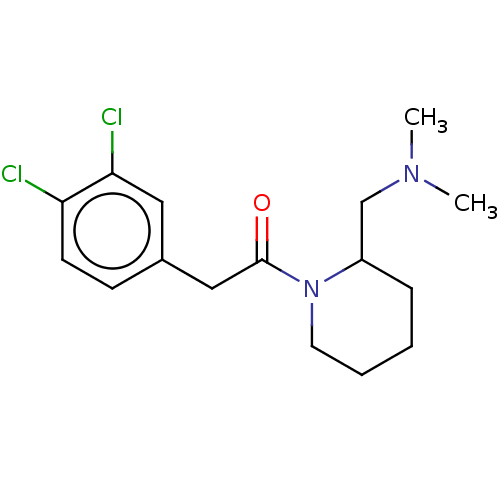
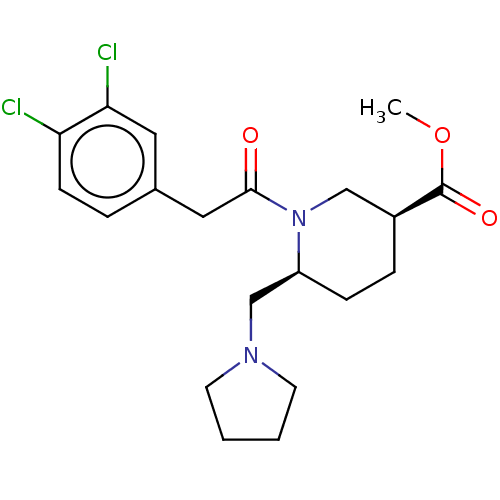
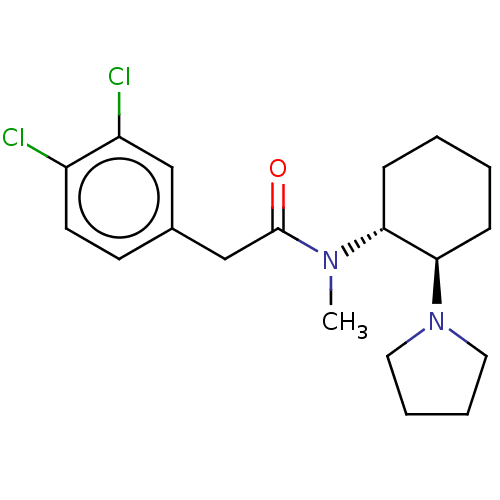
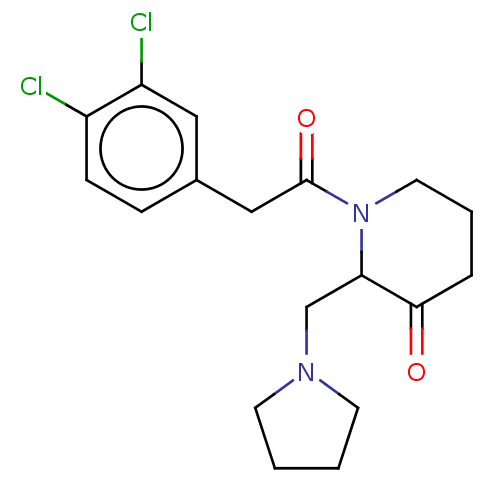
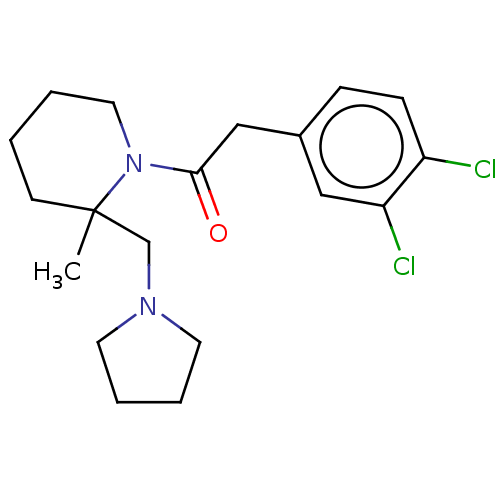
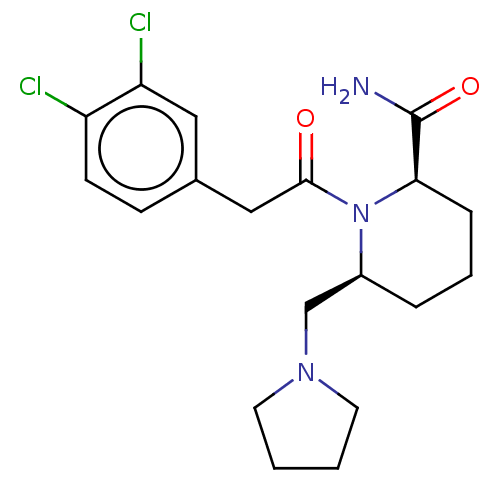
Affinity DataIC50: 0.100nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 0.120nMAssay Description:Opioid receptor kappa 1 agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Opioid receptor kappa 1 agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 0.880nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 3.5nMAssay Description:Opioid receptor kappa 1 agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 4.70nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 6.30nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Kappa-opioid receptor agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 29nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 46nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 57nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 69nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 102nMAssay Description:Opioid receptor kappa 1 agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 120nMAssay Description:Opioid receptor kappa 1 agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 160nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 250nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 260nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 370nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 390nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 550nMAssay Description:Opioid receptor kappa 1 agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 590nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 690nMAssay Description:Opioid receptor kappa 1 agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 970nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 1.02E+3nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+3nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: 2.20E+3nMAssay Description:Opioid receptor kappa agonist potency of the compound was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Opioid receptor kappa agonist potency was determined in vitro using rabbit vas deferens(LVD) preparationMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Opioid receptor kappa 1 agonist potency was determined in vitro using rabbit vas deferens (LVD) preparationMore data for this Ligand-Target Pair